Response rates to direct antiviral agents among hepatitis C virus infected patients who develop hepatocellular carcinoma following direct antiviral agents treatment
Abstract
Aim: Patients with chronic hepatitis C virus (HCV) infection who develop hepatocellular carcinoma (HCC) soon after treatment with direct antiviral agents (DAA) may have been harboring hitherto hidden tumors. If this were true, they should have a lower sustained viral response (SVR) rate, since active HCC hampers DAA efficacy. We aimed to verify this hypothesis.
Methods: We included all patients who attended an HCV clinic, provided that they: (1) had no previous history of HCC; (2) had received at least one DAA dose; and (3) had been followed-up clinically and ultrasonographically for at least six months after concluding DAA.
Results: The study population included n = 789 patients (55% males, median age 62 years). A median of 9.3 months (8.8-11.9) after concluding DAA, n = 19 (2.4%) patients were discovered to harbor HCC. In comparison to all others, patients with HCC were more commonly male (84% vs. 54%, P = 0.009), obese (47% vs. 17%, P = 0.002), and cirrhotic (95% vs. 35%, P < 0.001) and had less commonly achieved an SVR (68% vs. 98%, P < 0.001). Moreover, they had a trend for being less commonly treatment naïve (58% vs. 67%, P = 0.051). Based on multivariate analysis, the independent predictors of HCC were male sex (P = 0.031), cirrhosis (P = 0.004), obesity (P = 0.006), and failure to achieve an SVR (P < 0.001).
Conclusion: Lack of achieving SVR is a strong independent predictor of development of HCC early after treatment of hepatitis C with DAA. Treatment failure should further alert clinicians to the possibility of this dreadful complication.
Keywords
Introduction
Clearance of hepatitis C virus (HCV) infection, a major health problem, is obtainable by treating infected patients with one of several combinations of direct antiviral agents (DAA)[1]. Today, this desirable outcome can be reached so predictably and safely that HCV eradication is considered by many an achievable goal both on a local scale and on a global scale. Indeed, in 2016, the World Health Organization launched a campaign that - if successful - would eliminate viral hepatitis as a major threat to global health, with substantial economic benefits. Most importantly, putting HCV infection under control would prevent over 1.2 million deaths annually[2]. For sure, DAA treatment allows curing HCV infection in patients with advanced liver disease, including those who had undergone curative treatments for hepatocellular carcinoma (HCC).
Soon after DAA were introduced in practice, however, surprisingly high HCC incidence and/or recurrence rates were reported, an observation that generated alarm and dismay among clinicians[3]. In fact, among HCV-related complications, HCC is the most fearsome; furthermore, in the last few years, its incidence appears to be increasing[4]. Doubts that viral clearance by DAA might favor emergence of HCC clones by reducing immune pressure on HCV have been dispelled[2]: In fact, recent studies demonstrate convincingly that the opposite is true, i.e., DAA-induced sustained viral response (SVR) reduces the risk for de novo HCC[5-7]. The current interpretation is that the controversy - which has had the merit of highlighting the need to continue HCC surveillance in patients with cirrhosis, despite their achievement of an SVR - might have been generated mainly by inconsistencies and methodological limitations that flawed earlier studies[8].
Conceivably, among patients “cured” of HCC, so-called “recurrences” may actually represent prevalent tumors, whose presence is recognized only after DAA treatment is started. Could the same explanation apply to the apparent increase of HCC de novo diagnosed after DAA treatment? By definition, the presence of HCC foci should have been excluded to call these HCCs de novo; however, surveillance of HCC relies on ultrasonography, whose sensitivity is limited. A clue - if not definitive proof - in favor of the hypothesis that hidden HCC foci might have already been present when DAA were started would be to observe lower than expected SVR among DAA-treated patients later found to have an incident HCC, since patients with active HCC respond sub-optimally to DAA[9]. In the present study, we aimed to substantiate this hypothesis.
Methods
Patients
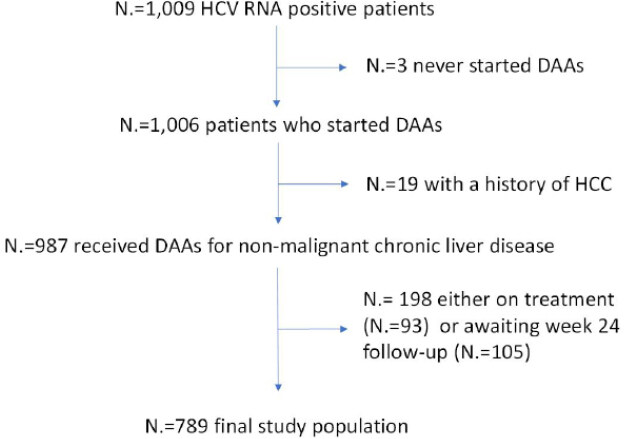
The study population included a cohort of consecutively recruited patients attending an academic liver clinic in Northern Italy to receive interferon-free treatment for chronic hepatitis in accordance to the European Association for Liver Diseases (EASL) guidelines[10]. Inclusion criteria were: (1) no previous diagnosis of HCC; and (2) minimum follow-up after the end of treatment of 180 days. Figure 1 presents the flow chart of the study.
Figure 1. Flow chart of the study. HCV: chronic hepatitis C virus; DAA: direct antiviral agents; HCC: hepatocellular carcinoma
The main demographic and clinical features of the patients enrolled are presented in Table 1.
Main characteristics of the study population
| Variable | n = 789 |
| Age, years | 62 (52-74) |
| Male:Female, n | 431(55):358(45) |
| Caucasian race, n | 762(97) |
| Body mass indexA, kg/m2 ≥ 30 kg/m2 | 25.1 (22.6-28.4)
136(18) |
| Diabetes, n | 112(14) |
| Prediabetes, n | 156(20) |
| Cirrhosis, n | 284(36) |
| HCV Genotype, n HCV-1A HCV-1B HCV-2 HCV-3 HCV-4 HCV-5 HCV-6 Undetermined | 115(15)
307(39) 205(26) 102(13) 55(7) 1 (< 1) 1 (< 1) 3 (< 1) |
| HCV RNA, UI/mL (× 1000)
< 400 400-4000 > 4000 | 994 (276-2280)
236(30) 462(59) 91(12) |
| Viral coinfections, n None HIV HBV (included 1 HDV positive) | 733(93)
46(6) 10(1) |
| Treatment history, n Naïve Experienced, interferon based regimens Experienced, direct antiviral agents | 530(67)
247(31) 12(2) |
The direct acting antiviral agent regimens used were sofosbuvir-based in n = 370 cases (47%) and protease inhibitor-based in 419 cases (53%). Among the n = 12 patients who were DAA-experienced, n = 6 (50%) had previously failed at least one interferon-based regimen.
Disease stage was assessed in all patients either invasively, with liver biopsy (n = 22, 2.8%), or non-invasively, with transient elastography (Fibroscan®; n = 744, 94.3%); in further few cases, a clinical diagnosis of liver cirrhosis was made (n = 23, 2.9%). Liver fibrosis in biopsies was staged from F0 to F4 according to the METAVIR staging system[11]. A liver stiffness threshold of 12.5 kPa was indicative of cirrhosis[12]. All patients were screened with ultrasound before starting antiviral treatment, irrespective of the presence of cirrhosis.
A clinical diagnosis of cirrhosis was reached in the presence of signs of liver decompensation and/or portal hypertension. HCC was diagnosed according to current EASL guidelines, which require a computed tomography (CT) scan or dynamic contrast-enhanced magnetic resonance imaging (MRI), showing typical hallmarks (hypervascularity in the arterial phase followed by washout in the portal or delayed phases). Focal lesions without typical hallmarks of HCC and those that developed in the absence of cirrhosis were subjected to a liver biopsy and confirmation by an expert liver pathologist[13].
The outcomes of antiviral therapy were defined as follows:
SVR: HCV RNA undetectable by a sensitive real-time polymerase chain reaction (PCR)-based assay, performed either after 12 or 24 weeks after the end of treatment;
relapse: presence of detectable HCV RNA at either post-treatment week 12 or post-treatment week 24, having HCV RNA found undetectable at the end of treatment;
dropout: patients who did not complete treatment as scheduled;
lost to follow-up: patients who did not perform a Week 12 or Week 24 after the end of treatment visit, although they completed treatment as scheduled.
Virological methods
Circulating HCV Ribonucleic Acid (HCV-RNA) was searched with the diagnostic system of Abbott RealTime HCV (Abbott, Wiesbaden, Germany), which has a sensitivity cut-off of 12 UI/mL; and the genotyping was performed by means of Abbott RealTime HCV Genotype II (Abbott).
Statistical analysis
Statistical analysis was performed using Stata Rel. 15.1 (StataCorp LLC, College Station TX, USA). As for continuous variables, the measures of centrality and dispersion of data were median and interquartile range, respectively, while comparisons between groups were carried out by the Mann-Whitney test. With regard to categorical variables, data are presented as frequencies (%), while the associations between groups were verified by the Fisher’s exact test or the Pearson chi square test, as appropriate. Logistic regression analysis was conducted to identify predictor(s) of de novo HCC among a set of independent variables. The threshold for statistical significance was 0.05 (two tails) for all tests used.
Results
Virologic outcomes
When analyzed with an intention-to-treat approach, SVR was 770/789 (97.6%). In detail, among the 19 patients who did not reach a SVR, 14/19 (74%) patients had a relapse, while 5/19 (26%) did not complete treatment (n = 1), died before reaching the 12 week post-treatment (n = 2), or performed neither the Post-Treatment Week 12 nor the Post-Treatment Week 24 visits (n = 2). Thus, the rate of virologic failure in this study was 1.8%. SVR was similar in patients who received a sofosbuvir-based regimen (359/370, 97%) vs. patients who received a protease inhibitor-based regimen (411/419, 98%) (P = 0.360), and in HCV-3 infected (98/102, 96%) vs. non-HCV-3 infected patients (672/688, 98%) (P = 0.312). There was a non-significant trend for lower SVR in patients with cirrhosis (273/284, 96%) vs. non-cirrhotic patients (497/505, 98%) (P = 0.054).
Development of de novo HCC
Along a median follow-up of 9.3 (interquartile range, 8.8-11.9) months, n = 19/789 (2.4%) patients were discovered to harbor HCC. The diagnosis was based on radiological criteria in 18/19 of patients (95%). Table 2 presents the main characteristics of these patients in comparison to all other patients.
Comparison between patients with and without de novo HCC at the end of follow-up
| Variable | HCC de novo n = 19 (2.4%) | No HCC n = 770 (97.6%) | P |
|---|---|---|---|
| Age, years | 68 (58-73) | 62 (52-74) | 0.248 |
| Male sex, n | 16(84) | 415(54) | 0.009 |
| Caucasian race, n | 19(100) | 743(96)A | 1.000 |
| Body mass indexA, kg/m2 ≥ 30 kg/m2 | 29.4 (21.3-31.6)
9(47) | 25.1 (22.6-28.3)
127/752(17) | 0.060
0.002 |
| Either diabetes or prediabetes, n | 9(47) | 259(34) | 0.226 |
| Cirrhosis, n | 18(95) | 266(35) | < 0.001 |
| HCV-3 Genotype, n | 2(11) | 100(13) | 1.000 |
| HCV RNA, UI/mL (× 1000)
> 4000 | 895 (255-1700)
2(11) | 1001 (276-2281)
89(12) | 0.763
1.000 |
| Coinfected, n | 0 (0) | 56(7) | 0.389 |
| Treatment history, n Naïve Experienced, IFN and/or DAA | 6(32)
13(68) | 524(68)
246(32) | 0.002 |
| SVR, n | 13(68) | 757(98) | < 0.001 |
Among patients who developed de novo HCC after antiviral therapy, 15/19 (79%) had either one or two nodules at the diagnosis, 3/19 (16%) had three or more nodules, and one patient had a diffuse infiltrative pattern (5%). Moreover, 7/19 (37%) had portal vein thrombosis (including complete or partial and segmental or sub-segmental thrombosis). Twelve patients (63%) fulfilled Milan Criteria[14].
Based on multivariate analysis, conducted having de novo HCC as dependent variable and age, male sex, obesity, cirrhosis, previous treatment history, and SVR as independent variables, the only independent predictors were male sex, obesity, cirrhosis, and SVR. The logistic regression model is summarized in Table 3.
Multivariate analysis of factors associated with de novo HCC development at the end of follow-up
| Variable | Odds ratio | 95%CI | P |
|---|---|---|---|
| Age | 1.03 | 0.99-1.08 | 0.131 |
| Male sex | 4.45 | 1.14-17.3 | 0.031 |
| Obesity | 4.68 | 1.55-14.1 | 0.006 |
| Cirrhosis | 21.1 | 2.68-166.1 | 0.004 |
| Treatment-experienced | 1.61 | 0.52-5.02 | 0.410 |
| Failure to achieve SVR | 24.2 | 5.76-101.8 | < 0.001 |
Discussion
The present study documented that, in the experience of a single center, the strongest predictor of HCC development following treatment of HCV infection was the lack of achieving SVR; other important pre-treatment factors were presence of cirrhosis, male gender, and obesity. These data confirm findings in other clinical and experimental studies, but they also have some novel practical implications that, in our opinion, may be worth considering.
It is well known that male gender represents a risk factor to develop HCC[15], although the reasons for the strong gender difference in HCC remain unclear. In the Italian population, the male to female ratio of HCC from any cause is 2.2 to 1, similar to what is observed in other western countries. In our cohort, the male to female ratio was higher, 5.3 to 1, possibly reflecting in part the age-specific sex difference in the incidence of HCC, which peaks at a slightly younger age than the one we observed in our study population[16]. The highest incidence of HCC in men could be related to the higher prevalence of cirrhosis in males due to more rapid disease progression before age 50 years. In fact, women during their reproductive years have a better control of HCV replication, possibly due to estrogens, and this fact leads to less necroinflammatory response and less fibrosis progression[17]. Others have suggested that estrogens have a direct putative antifibrogenic activity, or an interference with metabolic parameters and oxidative stress[18-20]. Finally, higher, genetically determined expression of interleukin-6 in males may also be a factor[21].
Most experts would agree that cirrhosis of any etiology is the strongest predictor of HCC. In fact, cirrhosis can be considered a premalignant condition, independently from the underlying liver disease[22-24]. It is worth mentioning that we staged liver disease mainly by transient elastography; while this is consistent with what is recommended by current European guidelines on hepatitis C[1], consideration must be given to the fact that the performance of this test may be suboptimal in obese patients. Hepatocarcinogenesis represents a multistep process, leading to chronic liver damage through persistent inflammatory damage that promotes malignant transformation[25-27]. The annual risk of HCC is as high as 3% in patients with cirrhosis and active HCV infection[28]. Viral hepatocarcinogenesis can be due to direct or indirect mechanisms, and is affected by host and environmental factors, such as alcohol intake, smoking, and HBV or HIV co-infections, which also increase the risk of cirrhosis. Indeed, although the estimated risk of HCC is increased 15-20-fold among persons infected with HCV in comparison to those who are not infected, most of the excess risk is limited to those with advanced hepatic fibrosis or cirrhosis[29].
In the present study, obesity was a major independent predictor of HCC. This observation is fully consistent with current literature that suggests the existence of a vicious circle linking cirrhosis/fibrosis, HCV infection, and lipid metabolism derangement. In the obese, the inactivation of negative regulators of STAT-1 and STAT-3 signaling drives the development of non-alcoholic steatohepatitis and HCC, not only in cirrhotic patients, but also in patients with chronic hepatitis[30,31]. Moreover, there is evidence that HCV-infected patients are prone to develop features of metabolic syndrome (MetS), probably due to the fact that the replication cycle of HCV depends heavily on the pathways of lipid metabolism in hepatocytes and considerably alters host lipid hemostasis[32,33]. Interestingly, two large population cohort studies from Taiwan showed that HCV infection was strongly associated with MetS. The prevalence of MetS in these patients ranged from 13% to 32%; they had an aggressive and severe liver disease, developing more severe fibrosis than those without MetS, which contributed to cancer development[34-36]. However, MetS did not affect SVR achievement after DAA[37].
The major novelty of the present study lies in the strong association observed between lack of SVR and identification of HCC soon after concluding DAA. Overall, the rates of SVR and virologic failure in our cohort (97.6% and 1.8%, respectively) were comparable with what has been observed in registration trials[38-40] and in real-life cohorts[25,41,42]. While some authors affirmed in several retrospective studies that DAA increased the rate of early recurrence/occurrence of HCC, the short follow-up, the small number of patients, and the study design did not allow definite conclusions[3,43,44]. In contrast, multiple large cohort studies and meta-analyses have since demonstrated that DAA-induced SVR is associated with reduced risk of HCC occurrence[45-49].
One possible explanation for the low SVR in those who develop HCC is that it derives from the sum of risk factors for HCC, such as older age, high alcohol intake, more severe fibrosis, and co-infections[50-53]. Although the difference did not reach statistical significance, we did observe a numerically lower SVR rate in cirrhotic vs. non-cirrhotic patients, which - combined with the low number of cirrhotic patients who develop HCC - may create a bias. However, the results logistic regression analysis strongly support the independence of these two variables in predicting the development of HCC (it should also be noted that the test is not designed to compare the relative strength of each variable in the model).
A different way to reconcile these findings is to hypothesize that the apparent increase of HCC incidence/recurrence rates might be due to the difficulties of identifying small HCC foci by current screening methods. Not surprisingly, compared to explant pathology, ultrasound is insufficiently sensitive in detecting HCC in obese patients[54] and obesity hampers the quality of HCC surveillance[55-57]. Thus, one may speculate that, especially in obese patients, what is observed as de novo HCC is in fact missed HCC, hence the low SVR rate among those who “develop” HCC in our cohort. Most of the DAA failures in patients with previous HCC diagnosis occurred among patients with active cancers, where DAA failed in almost half of the cases[9], possibly because HCC may serve as a sanctuary for HCV. In agreement with these findings, in a preliminary report from our group, we did observe unusually low SVR rates among de novo and recurrent HCC cases, leading us to suggest that treatment failure should be considered a clue of a yet undetected HCC[58,59].
We must acknowledge several limitations of our work. First, it is a single-center study, with a short follow-up, during which - luckily enough - only a relatively small number of patients went on to develop HCC. Being a retrospective analysis of data generated in clinical practice, we screened our patients before and after DAA treatment with ultrasound, thus we are unable to provide pre-treatment data on higher level dynamic imaging (CT or MRI), which was performed only in the presence of suspicious focal liver lesions. Finally, we do not have reliable data about current and past alcohol intake in our study population, which are traditionally quite difficult to obtain. Nevertheless, at least in our opinion, the study conveys two messages worth considering: (1) given the extremely high SVR rates obtainable today in all subgroups of HCV infected patients, when DAA treatment fails, the possibility that the patient harbors HCC should come to mind; and (2) in male, obese, cirrhotic HCV-infected patients, a second level imaging technique should confirm that they are free of HCC before starting a DAA regimen.
In conclusion, the present study indicates virologic failure as a strong independent predictor for de novo HCC identification early after treatment of hepatitis C with DAA. Clearly, all patients with cirrhosis regardless of SVR response should be monitored at regular six-month intervals, since cirrhosis - either in the presence or in the absence of HCV - is the dominant risk factor for HCC. However, lack of achieving SVR should further alert clinicians to the possibility of this dreadful complication, especially among HCV carriers who are male, obese, and cirrhotic.
Declarations
Authors’ contributionsConceptualization, data curation, formal analysis, supervision, investigation, validation, visualization, writing - original draft, writing - review & editing: Burlone ME
Conceptualization, data curation, formal analysis, supervision, validation, visualization, writing -review & editing: Fangazio S
Data curation, investigation, validation, visualization: Croce A, Ceriani E, Rapetti R, Rigamonti C, Smirne C
Data curation, investigation, supervision, validation, visualization: Tonello S
Supervision, validation, visualization: Ravanini P
Data curation, investigation, supervision, validation, visualization: Minisini R
Conceptualization, data curation, formal analysis, investigation, methodology, project administration, supervision, validation, visualization, writing - review & editing: Pirisi M
Availability of data and materialsRaw data are available upon request.
Financial support and sponsorshipNone.
Conflicts of interestAll authors declared that there are no conflicts of interest.
Ethical approval and consent to participateBeing a retrospective analysis of anonymized data of patients treated in standard clinical practice, no ethical committee approval was required. However, all patients gave written informed consent for their participation to the study, which was conducted in strict accordance to the Principles of the Declaration of Helsinki.
Consent for publicationNot applicable.
Copyright© The Author(s) 2020.
REFERENCES
1. Pawlotsky JM, Negro F, Aghemo A, Berenguer M, Dalgard O, et al. EASL recommendations on treatment of hepatitis C 2018. J Hepatol 2018;69:461-511.
2. World Health Organization. Global hepatitis report, 2017. 2017.
3. Reig M, Mariño Z, Perelló C, Iñarrairaegui M, Ribeiro A, et al. Unexpected high rate of early tumor recurrence in patients with HCV-related HCC undergoing interferon-free therapy. J Hepatol 2016;65:719-26.
4. Akinyemiju T, Abera S, Ahmed M, Alam N, Alemayho MA, et al. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level results from the global burden of disease study 2015. JAMA Oncol 2017;3:1683-91.
5. Mashiba T, Joko K, Kurosaki M, Ochi H, Osaki Y, et al. Does interferon-free direct-acting antiviral therapy for hepatitis c after curative treatment for hepatocellular carcinoma lead to unexpected recurrences of hcc? A multicenter study by the Japanese red cross hospital liver study group. PLoS One 2018;13:e0194704.
6. Nagata H, Nakagawa M, Asahina Y, Sato A, Asano Y, et al. Effect of interferon-based and -free therapy on early occurrence and recurrence of hepatocellular carcinoma in chronic hepatitis C. J Hepatol 2017;67:933-9.
7. Virlogeux V, Pradat P, Hartig-Lavie K, Bailly F, Maynard M, et al. Direct-acting antiviral therapy decreases hepatocellular carcinoma recurrence rate in cirrhotic patients with chronic hepatitis C. Liver Int 2017;37:1122-7.
8. Su F, Ioannou GN. Hepatocellular carcinoma risk after direct-acting antiviral therapy. Clin Liver Dis 2019;13:6-12.
9. Prenner SB, VanWagner LB, Flamm SL, Salem R, Lewandowski RJ, et al. Hepatocellular carcinoma decreases the chance of successful hepatitis C virus therapy with direct-acting antivirals. J Hepatol 2017;66:1173-81.
10. European Association for the Study of the Liver. EASL recommendations on treatment of hepatitis C 2016. J Hepatol 2017;66:153-94.
11. Bedossa P. Intraobserver and interobserver variations in liver biopsy interpretation in patients with chronic hepatitis C. Hepatology 1994;20:15-20.
12. Castera L, Forns X, Alberti A. Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol 2008;48:835-47.
13. Galle PR, Forner A, Llovet JM, Mazzaferro V, Piscaglia F, et al. EASL clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 2018;69:182-236.
14. Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 1996;334:693-700.
15. Jemal A, Bray F, Center MM, Ferlay J, Ward E, et al. Global cancer statistics. CA Cancer J Clin 2011;61:69-90.
16. Liu P, Xie SH, Hu S, Cheng X, Gao T, et al. Age-specific sex difference in the incidence of hepatocellular carcinoma in the United States. Oncotarget 2017;8:68131-7.
17. Burlone ME, Pedrinelli AR, Giarda P, Minisini R, Pirisi M. Influence of age on sex-related differences among patients with hepatitis C. Eur J Gastroenterol Hepatol 2016;28:1100-1.
18. Davis GL, Alter MJ, El-Serag H, Poynard T, Jennings LW. Aging of hepatitis C virus (HCV)-infected persons in the United States: a multiple cohort model of HCV prevalence and disease progression. Gastroenterology 2010;138:513-21.
19. Nasta P. “Immune activation, aging and gender” and progression of liver disease. Acta Biomed 2011;82:115-23.
20. Prieto J. Inflammation, HCC and sex: IL-6 in the centre of the triangle. J Hepatol 2008;48:380-1.
21. Cussigh A, Falleti E, Fabris C, Bitetto D, Cmet S, et al. Interleukin 6 promoter polymorphisms influence the outcome of chronic hepatitis C. Immunogenetics 2011;63:33-41.
22. Fattovich G, Stroffolini T, Zagni I, Donato F. Hepatocellular carcinoma in cirrhosis: incidence and risk factors. Gastroenterology 2004;127 (5 Suppl 1):S35-50.
23. Simonetti RG, Cammà C, Fiorello F, Politi F, D’Amico G, et al. Hepatocellular carcinoma - a worldwide problem and the major risk factors. Dig Dis Sci 1991;36:962-72.
24. Tsukuma H, Hiyama T, Tanaka S, Nakao M, Yabuuchi T, et al. Risk factors for hepatocellular carcinoma among patients with chronic liver disease. N Engl J Med 1993;328:1797-1801.
25. Akinyemiju T, Abera S, Ahmed M, Alam N, Alemayohu MA, et al. The burden of primary liver cancer and underlying etiologies from 1990 to 2015 at the global, regional, and national level. JAMA Oncol 2017;3:1683-91.
26. Degos F, Christidis C, Ganne-Carrie N, Farmachidi JP, Degott C, et al. Hepatitis C virus related cirrhosis: time to occurrence of hepatocellular carcinoma and death. Gut 2000;47:131-6.
27. West J, Card TR, Aithal GP, Fleming KM. Risk of hepatocellular carcinoma among individuals with different aetiologies of cirrhosis: a population-based cohort study. Aliment Pharmacol Ther 2017;45:983-90.
29. Fabris C, Smirne C, Toniutto P, Colletta C, Rapetti R, et al. Assessment of liver fibrosis progression in patients with chronic hepatitis C and normal alanine aminotransferase values: the role of AST to the platelet ratio index. Clin Biochem 2006;39:339-43.
30. Alexander J, Torbenson M, Wu TT, Yeh MM. Non-alcoholic fatty liver disease contributes to hepatocarcinogenesis in non-cirrhotic liver: a clinical and pathological study. J Gastroenterol Hepatol 2013;28:848-54.
31. Grohmann M, Wiede F, Dodd GT, Gurzov EN, Ooi GJ, et al. Obesity drives STAT-1-dependent NASH and STAT-3-dependent HCC. Cell 2018;175:1289-306.
32. Burlone ME, Budkowska A. Hepatitis C virus cell entry: role of lipoproteins and cellular receptors. J Gen Virol 2009;90:1055-70.
33. Schaefer EA, Chung RT. HCV and host lipids: an intimate connection. Semin Liver Dis 2013;33:358-68.
34. Kuo YH, Kee KM, Wang JH, Hsu NT, Hsiao CC, et al. Association between chronic viral hepatitis and metabolic syndrome in southern Taiwan: a large population-based study. Aliment Pharmacol Ther 2018;48:993-1002.
35. Perz JF, Armstrong GL, Farrington LA, Hutin YJF, Bell BP. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol 2006;45:529-38.
36. Yoon H, Lee JG, Yoo JH, Son MS, Kim DY, et al. Effects of metabolic syndrome on fibrosis in chronic viral hepatitis. Gut Liver 2013;7:469-74.
37. Dong TS, Aby ES, Benhammou JN, Kawamoto J, Han SH, et al. Metabolic syndrome does not affect sustained virologic response of direct-acting antivirals while hepatitis C clearance improves hemoglobin A1c. World J Hepatol 2018;10:612-21.
38. Dore GJ, Conway B, Luo Y, Janczewska E, Knysz B, et al. Efficacy and safety of ombitasvir/paritaprevir/r and dasabuvir compared to IFN-containing regimens in genotype 1 HCV patients: the MALACHITE-I/II trials. J Hepatol 2016;64:19-28.
39. Zeuzem S, Foster GR, Wang S, Asatryan A, Gane E, et al. Glecaprevir-pibrentasvir for 8 or 12 weeks in HCV genotype 1 or 3 infection. N Engl J Med 2018;378:354-69.
40. Zeuzem S, Ghalib R, Reddy KR, Pockros PJ, Ari ZB, et al. Grazoprevir-Elbasvir combination therapy for treatment-naive cirrhotic and noncirrhotic patients with chronic hepatitis C virus genotype 1, 4, or 6 infection: a randomized trial. Ann Int Med 2015;163:1-13.
41. Buggisch P, Vermehren J, Mauss S, Günther R, Schott E, et al. Real-world effectiveness of 8-week treatment with ledipasvir/sofosbuvir in chronic hepatitis C. J Hepatol 2018;68:663-71.
42. Londoño MC, Garcia-Buey L, Mallolas J, Barril G, Ahumada A, et al. Effectiveness, safety/tolerability of OBV/PTV/r ± DSV in patients with HCV genotype 1 or 4 with/without HIV-1 co-infection, chronic kidney disease (CKD) stage IIIb/V and dialysis in Spanish clinical practice - preliminary data Vie-KinD study. J Hepatol 2017;66:S718-9.
43. Cardoso H, Vale AM, Rodrigues S, Gonçalves R, Albuquerque A, et al. High incidence of hepatocellular carcinoma following successful interferon-free antiviral therapy for hepatitis C associated cirrhosis. J Hepatol 2016;65:1070-1.
44. Conti F, Buonfiglioli F, Scuteri A, Crespi C, Bolondi L, et al. Early occurrence and recurrence of hepatocellular carcinoma in HCV-related cirrhosis treated with direct-acting antivirals. J Hepatol 2016;65:727-33.
45. ANRS collaborative study group on hepatocellular carcinoma. Lack of evidence of an effect of direct-acting antivirals on the recurrence of hepatocellular carcinoma: data from three ANRS cohorts. J Hepatol 2016;65:734-40.
46. Baumert TF, Berg T, Lim JK, Nelson DR. Status of direct-acting antiviral therapy for hepatitis C virus infection and remaining challenges. Gastroenterology 2019;156:431-45.
47. Ioannou GN, Green PK, Berry K. HCV eradication induced by direct-acting antiviral agents reduces the risk of hepatocellular carcinoma. J Hepatol 2018.
48. Li DK, Ren Y, Fierer DS, Rutledge S, Shaikh OS, et al. The short-term incidence of hepatocellular carcinoma is not increased after hepatitis C treatment with direct-acting antivirals: an ERCHIVES study. Hepatology 2018;67:2244-53.
49. Waziry R, Hajarizadeh B, Grebely J, Amin J, Law M, et al. Hepatocellular carcinoma risk following direct-acting antiviral HCV therapy: a systematic review, meta-analyses, and meta-regression. J Hepatol 2017;67:1204-12.
50. Bagella P, Fiore V, Caruana G, Madeddu G. Editorial - non AIDS-defining malignancies: a new epidemic in HIV-infected population for the upcoming decades? European review for medical and pharmacological sciences 2017. Eur Rev Med Pharmacol Sci 2017;21:4744-5.
51. Di Benedetto F, Tarantino G, Ercolani G, Baccarani U, Montalti R, et al. Multicenter Italian experience in liver transplantation for hepatocellular carcinoma in HIV-infected patients. Oncoligst 2013;18:592-9.
52. Yang JD, Altekruse SF, Nguyen MH, Gores GJ, Roberts LR. Impact of country of birth on age at the time of diagnosis of hepatocellular carcinoma in the United States. Cancer 2017;123:81-9.
53. Yang JD, Mohamed HA, Cvinar JL, Gores GJ, Roberts LR, et al. Diabetes mellitus heightens the risk of hepatocellular carcinoma except in patients with hepatitis C cirrhosis. Am J Gastroenterol 2016;111:1573-80.
54. Esfeh JM, Hajifathalian K, Ansari-Gilani K. Sensitivity of ultrasound in detecting hepatocellular carcinoma in obese patients compared to explant pathology as the gold standard. Clin Mol Hepatol 2019;26:54-9.
56. Simmons O, Fetzer DT, Yokoo T, Marrero JA, Yopp A, et al. Predictors of adequate ultrasound quality for hepatocellular carcinoma surveillance in patients with cirrhosis. Aliment Pharmacol Ther 2017;45:169-77.
57. Singal A, Volk ML, Waljee A, Salgia R, Higgins P, et al. Meta-analysis: surveillance with ultrasound for early-stage hepatocellular carcinoma in patients with cirrhosis. Aliment Pharmacol Ther 2009;30:37-47.
58. Fangazio S, Camatta D, Tran Minh M, Ceriani E, Minisini R, et al. Treatment failure after interferon-free treatment of hepatitis C as a clue of a yet undetected hepatocellular carcinoma. J Hepatol 2017; doi: 10.1016/j.jhep.2017.09.004.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Burlone ME, Fangazio S, Croce A, Ceriani E, Rapetti R, Rigamonti C, Smirne C, Tonello S, Ravanini P, Minisini R, Pirisi M. Response rates to direct antiviral agents among hepatitis C virus infected patients who develop hepatocellular carcinoma following direct antiviral agents treatment. Hepatoma Res 2020;6:3. http://dx.doi.org/10.20517/2394-5079.2019.37
AMA Style
Burlone ME, Fangazio S, Croce A, Ceriani E, Rapetti R, Rigamonti C, Smirne C, Tonello S, Ravanini P, Minisini R, Pirisi M. Response rates to direct antiviral agents among hepatitis C virus infected patients who develop hepatocellular carcinoma following direct antiviral agents treatment. Hepatoma Research. 2020; 6: 3. http://dx.doi.org/10.20517/2394-5079.2019.37
Chicago/Turabian Style
Burlone, Michela Emma, Stefano Fangazio, Alessandro Croce, Elisa Ceriani, Rachele Rapetti, Cristina Rigamonti, Carlo Smirne, Stelvio Tonello, Paolo Ravanini, Rosalba Minisini, Mario Pirisi. 2020. "Response rates to direct antiviral agents among hepatitis C virus infected patients who develop hepatocellular carcinoma following direct antiviral agents treatment" Hepatoma Research. 6: 3. http://dx.doi.org/10.20517/2394-5079.2019.37
ACS Style
Burlone, ME.; Fangazio S.; Croce A.; Ceriani E.; Rapetti R.; Rigamonti C.; Smirne C.; Tonello S.; Ravanini P.; Minisini R.; Pirisi M. Response rates to direct antiviral agents among hepatitis C virus infected patients who develop hepatocellular carcinoma following direct antiviral agents treatment. Hepatoma. Res. 2020, 6, 3. http://dx.doi.org/10.20517/2394-5079.2019.37
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 7 clicks
Cite This Article 7 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.