Alpha-fetoprotein as a predictor of hepatocellular carcinoma recurrence following liver transplantation
Abstract
Alpha-fetoprotein (AFP) has been increasingly recognised as a valuable marker in predicting HCC recurrence post-liver transplantation. Moreover, its secretion has been associated with poor histological tumour characteristics as it reflects an aggressive tumour biological behaviour. This review aims to summarise the emerging evidence on the use of AFP either as an independent marker, or as a variable incorporated into prognostic models. For this purpose, an electronic PubMed literature search was performed. Due to the heterogeneity of the reported studies, drawing clear conclusions about the optimum AFP cut-off level to predict recurrence is difficult. Models that include AFP at different cut-offs have been shown be superior to Milan criteria in predicting disease recurrence, but need to be prospectively validated in order to confirm their prognostic value. Until more refined methods for selecting patients become available, existing evidence support the use of AFP in decision models for liver transplantation.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is expected to become a leading cause for liver transplantation (LT) following curative treatment for hepatitis C and after more widespread acceptance of the practice of tumour down-staging for patients originally considered beyond LT criteria[1]. It has been estimated that patients with HCC currently represent 30%-35% of the waiting list population in Europe and in an era of organ shortage, selecting the best candidates for LT with the lowest risk of post-transplant recurrence poses a clinical challenge[2].
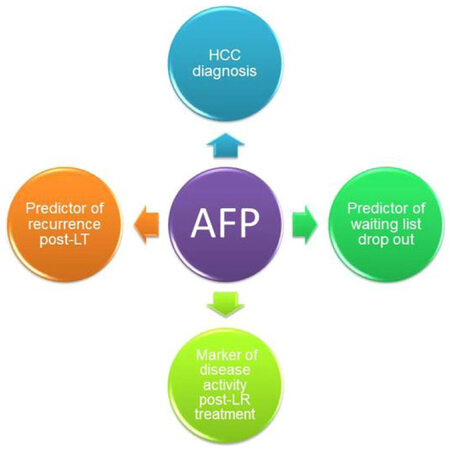
It is evident that even after applying the most restrictive tumour burden selection criteria, 10%-15% of patients with HCC still develop HCC recurrence post-LT[3]. It is also apparent that factors beyond tumour size and number are associated with more aggressive tumour biology and are accountable for increasing the risk of HCC recurrence[4]. Among these, alpha-fetoprotein (AFP) has been increasingly recognised as a valuable marker in predicting HCC recurrence [Figure 1][5]. Although an association between high AFP values and poor outcomes post LT has been established[6], it has still not been incorporated in the most widely used listing criteria. Several scoring systems encompassing pre-LT serum AFP levels and tumour size criteria have been recently proposed and have shown to have better predictability of recurrence compared to traditional Milan criteria[7-10].
Figure 1. The predictive role of AFP pre- and post-LT. AFP: alpha-fetoprotein; HCC: hepatocellular carcinoma; LT: liver transplantation
The aim of this review is to summarise the emerging evidence on the role of AFP as a predictor of HCC recurrence following LT, either as an independent value or after being combined with tumour size criteria into prognostic models. For this purpose, an electronic PubMed literature search was performed from January 2004 to April 2018 by using the following keywords: “alpha-fetoprotein”, “hepatocellular carcinoma”, “liver transplantation”, “recurrence” and “survival”. In this review, we have included all the original studies that evaluated the role of alpha-fetoprotein (AFP) or AFP including models as a predictive marker of prognosis following LT.
AFP as an independent predictor of recurrence and survival
Several studies have been published with regard to an AFP absolute cut-off value or AFP change on the waiting list, as an independent predictor of prognosis[5,9,11-13]. In a meta-analysis that included 13 studies[14], the upper cut-off AFP value varied widely between 20 ng/mL and 1000 ng/mL. Due to this variation between different studies, this meta-analysis was unable to suggest a single cut-off level which could be universally approved across centres. Despite that, a significant correlation between AFP and both post-transplant recurrence and prognosis was established in the majority of the reported studies[14].
The critical question as to whether downstaging HCC patients with high AFP is feasible and is associated with similar prognostic outcomes was addressed in a study by Merani et al.[6]. This retrospective analysis included 6817 patients that were listed with a diagnosis of HCC in the Scientific Registry of Transplant Recipients (SRTR) in the United States and showed that downstaging to an AFP level ≤ 400 ng/mL was associated with good survival rates and prognosis, regardless of the initial AFP level. More specifically, patients who were successfully downstaged to AFP ≤ 400 ng/mL had a similar dropout rate (10% in both groups) and post-transplant survival rates (89% vs. 78% at 3 years, P = 0.11) to patients with AFP levels persistently ≤ 400 ng/mL.
A strong dose-response relationship between AFP level and post-transplant outcomes was demonstrated in a study that utilised data from the UNOS registry, that included patients (n = 45,267) who were transplanted in the US between 2002 and 2011[15]. Although patients with an AFP < 15 ng/mL prior to transplantation had similar survival rates to patients without HCC, there was a significant decrease in survival as AFP increased; 16-65 ng/mL [adjusted hazard ratio (AHR) = 1.38, 95% CI : 1.23-1.54], 66-320 ng/mL (AHR = 1.65, 95% CI : 1.45-1.88), and greater than 320 ng/mL (AHR = 2.37, 95% CI : 2.06-2.73). In addition, patients with tumours beyond the Milan criteria at listing had excellent post-transplant survival if serum AFP level was ≤ 15 ng/mL (AHR = 0.97, 95% CI : 0.66-1.43). This study also showed that downstaging of AFP following locoregional treatment was associated with improved post-transplant survival and prognosis[15].
In a prospective study by Lai et al.[12], mRESIST (modified Response Evaluation criteria in Solid Tumours) progression following locoregional treatment and AFP slope > 15 ng/mL/month, as defined by the difference between the initial and the last pre-LT AFP value divided by the time span between the two values, were independent risk factors of recurrence and survival. Patients within and beyond radiological Milan criteria had similar recurrence-free and overall survival rates if they had stable disease post locoregional treatment and/or an AFP slope < 15 ng/mL/month. Similarly, patients within the Milan criteria but with either progressive disease or AFP slope > 15 ng/mL/month were shown to have increased recurrence rates compared to patients within or beyond Milan criteria with no risk factors[12]. Another retrospective study from the same group, has shown that AFP > 400 ng/mL can result in an 8-fold increase in the risk of recurrence and a combination with a total tumour diameter < 8 cm can result comparable 5 year survival and recurrence rates[16].
The predictive value of AFP slope, rather than an AFP single value alone, was also examined in a study by Vibert et al.[17], which included 252 patients transplanted between 1985 and 2005, in a single centre. AFP progression, as defined by an increase greater than 15 ng/mL monthly, was significantly associated with reduced 5 year recurrence-free (47% vs. 74%, P = 0.01) and overall survival (54% vs. 77%, P = 0.02). In the multivariate analysis, progression of AFP was independently associated with recurrence-free and overall survival. Interestingly, all examined static values of AFP prior to transplantation were not correlated with overall or recurrence-free survival. AFP progression was also significantly associated with the presence of vascular invasion and poor histological differentiation, which suggests that it can be a valuable surrogate pre-operative marker of unfavourable histological findings[17].
Another study by Hameed et al.[11] has shown that setting a cut-off value of 1000 ng/mL as an exclusion criteria for transplantation, would have resulted in a 20% reduction in the rate of HCC recurrence at the cost of excluding only 4.7% of patients listed. Following this observation, our own national (United Kingdom) Transplant guidelines have applied this cut-off level as an exclusion criterion for LT. This study has also demonstrated a strong correlation between AFP and micro-vascular invasion, especially in patients with AFP values varying between 300-1000 ng/mL[11].
A large retrospective European multicentre study intended to identify variables for selecting patients that would have the best benefit from transplantation[18]. AFP ≥ 1000 ng/mL, MELD ≤ 13, mRESIST progressive or complete response and within Milan criteria were associated with a poor intention to treat (ITT) benefit from LT. Based on these risk factors, four benefit groups were identified. Patients that met three out of four risk factors were shown to have no benefit from LT, and that accounted for 19.2% of the study population. This study has introduced the concept of ITT benefit for transplantation that can stratify patients towards a more effective allocation system.
In a separate study that included patients registered at the SRTR database (n = 6478), a total tumour volume (TTV) ≥ 115 cm3 and AFP > 400 were independent predictors of survival[19]. After combining the two variables, with patients being beyond criteria if they had either TTV ≥ 115 cm3 or an AFP > 400, the composite score efficiently predicted overall survival (HR 2, 95% CI: 1.7-2.4, P < 0.001).
A prospective validation of the proposed criteria was subsequently performed on 233 patients transplanted in 3 different centres. Patients with an AFP > 400 ng/mL and TTV > 115 cm3 were excluded from LT. Although the risk of drop out was higher in patients within TTV/AFP but beyond Milan criteria, a similar recurrence (9.4 vs. 4.4, P > 0.05) and survival rate (74.6% vs. 78.7%, P > 0.05) was demonstrated between the two groups. To account for the higher drop out and worse ITT survival in the TTV/AFP groups, the proposed listing criteria are recommended to centres with waiting time of 8 months or more[9].
A retrospective single centre study that included 137 recipients with more than 50% of the total patient number being beyond Milan or University of California San Francisco criteria at pre-transplant imaging, showed that tumour number > 3 based on explant findings, AFP level ≥ 400 ng/mL, microvascular invasion and rejection needing anti-lymphocytic antibodies were independent predictors of recurrence[20].
Another study from China has also confirmed that AFP ≥ 400 ng/mL is independently associated with adverse outcomes. Based on prognostic stratification, the Hangzhou criteria were proposed, based on which, patients with total tumour diameter less than or equal to 8 cm, or total tumour diameter more than 8 cm, with histopathologic grade I or II and preoperative AFP level less than or equal to 400 ng/mL simultaneously, were shown to have favourable post-transplant outcomes[21].
Several other studies have reported on different AFP values as predictors of recurrence in the recent literature. This includes a study that included 101 patients from a single centre in the U.S. showed that AFP > 100 ng/mL (OR = 5.0, 95% CI : 1.23-29.71, P = 0.006) and tumour size on explant (OR = 4.1, 95% CI : 1.2-13.5, P = 0.013) were associated with microvascular invasion and post-LT recurrence[22]. Another single centre study including 140 HCC patients confirmed the validity of AFP > 100 ng/mL as cut-off value in predicting the risk of post-LT recurrence, in patients meeting the San Francisco or up-to-seven criteria (Warsaw criteria)[23]. The authors have shown that the expanded proposed criteria increased the transplant eligibility rate by 20.3% without compromising post-transplant outcomes.
A multi-centre Korean study that included 688 patients with advanced HCC (beyond Milan criteria on explant) or far advanced HCC (defined as maximum tumour diameter ≥ 10 cm, 10 or more nodules, or accompanying macro-vascular invasion) has shown that both AFP and the biomarker prothrombin induced by vitamin K absence or antagonist-II (PIVKA-II) were significant risk factors for recurrence[24]. In particular, a sum of AFP plus PIVKA-II < 300 was a better predictor than either marker alone and can provide valuable information on tumour biology and behaviour in advanced HCCs.
Finally, a single centre study of 250 Korean patients has shown that patients with AFP > 400 ng/mL had significantly worse disease-free and overall survival[25]. On discriminative analysis; a cut off value of 54 ng/mL was significantly associated with disease recurrence, whereas cut-off value of 105 ng/mL was a better discriminator of overall survival[25].
Table 1 summarises the studies that have evaluated different cut-off values of AFP as significant predictors of recurrence and survival following LT.
AFP values to predict HCC recurrence in recently published studies
| Reference | Year of publication | Number of patients | Country | Study design | AFP Cut-off value | Prognostic endpoint |
|---|---|---|---|---|---|---|
| Duvoux et al.[10] | 2012 | 537 TC 435 VC | France | Retrospective Prospective | 100 ng/mL, 1000 ng/mL | 5-year RFS and OS |
| Mazzaferro et al.[7] | 2017 | 1018 TC 341 VC | Italy China | Retrospective Retrospective | 200 ng/mL,400 ng/mL 1000 ng/mL | 5-year OS |
| Mehta et al.[8] | 2017 | 721 TC 340 VC | US Canada | Retrospective Retrospective | 100 ng/mL, 1000 ng/mL | 5-year RFS and OS |
| Merani et al.[6] | 2011 | 6817 | US | Retrospective | 400 ng/mL | 3-year ITT survival and OS |
| Berry et al.[15] | 2013 | 45,267 | US | Retrospective | 15 ng/mL, 16-65 ng/mL 66-320 ng/mL, < 320 ng/mL | 6-year OS |
| Lai et al.[12] | 2013 | 422 | Europe | Prospective | AFP slope > 15 ng/mL/month | 5-year RFS and OS |
| Lai et al.[16] | 2012 | 158 | Italy | Retrospective | AFP > 400 ng/mL | 5-year RFS and OS |
| Vibert et al.[17] | 2010 | 252 | France | Retrospective | AFP slope > 15 ng/mL/month | 5-year RFS and OS |
| Hameed et al.[11] | 2014 | 211 | US | Retrospective | AFP > 1000 ng/mL | 1-, 5-year RFS and OS |
| Lai et al.[18] | 2017 | 2013 | Europe | Retrospective | AFP > 1000 ng/mL | ITT survival |
| Toso et al.[19] | 2009 | 6478 | US | Retrospective | AFP > 400 ng/mL | 5-year OS |
| Toso et al.[9] | 2015 | 233 | Switzerland Canada | Prospective | AFP > 400 ng/mL | 4-year RFS and OS, ITT survival |
| Ciccarelli et al.[20] | 2012 | 137 | Belgium | Retrospective | AFP > 400 ng/mL | 5-year RFS |
| Zheng et al.[21] | 2008 | 195 | China | Retrospective | AFP > 400 ng/mL | 1-,3-,5- year RFS and OS |
| McHugh et al.[22] | 2010 | 101 | US | Retrospective | AFP > 100 ng/mL | 1-,3-,5- year RFS and OS |
| Grat et al.[23] | 2017 | 140 | Poland | Retrospective | AFP > 100 ng/mL | 5-year RFS and OS |
| Lee et al.[24] | 2018 | 688 | Korea | Retrospective | AFP + PIVKA > 300 | 5-year RFS and OS |
| She et al.[25] | 2018 | 250 | Korea | Retrospective | 54 ng/mL 105 ng/mL | 5-year RFS 5-year OS |
AFP prognostic scores for the selection of patients for LT
In the recent literature, several models that combine tumour burden characteristics with pre-operative AFP at different cut-off levels have been proposed. These have been shown to be superior to Milan criteria in predicting tumour recurrence.
AFP score
A prognostic model which includes AFP at two different cut-off levels (100 ng/mL and 1000 ng/mL) and tumour radiological characteristics at listing was developed in a cohort of 597 French patients transplanted for HCC across 16 different centres, and prospectively validated in a cohort of 434 patients registered for LT in France[10]. The AFP score defined three groups of patients with low risk of HCC recurrence; (1) patients with 1-3 nodules, maximum diameter of the largest tumour of less than 3 cm and AFP ≤ 1000 ng/mL, (2) patients with 1-3 nodules, maximum diameter of the largest tumour of 3-6 cm, and AFP ≤ 100 ng/mL and (3) patients with more than 4 nodules, maximum diameter of the largest tumour of less than 3 cm, and AFP ≤ 100 ng/mL.
A simplified user-friendly version of the model was developed and the score was calculated by adding the individual points from each variable [Table 2]. A cut- off value more than two (2) points discriminated between patients with low and high risk of recurrence. Five-year recurrence rate was 8.8% ± 1.7% vs. 50.6% ± 10.2% (P < 0.001) in patients with AFP score ≤ 2 and ≥ 2 and 5-year survival rate was 67.8% ± 3.4% and 47.5% ± 8.1% (P < 0.002) respectively[10].
The AFP score for the prediction of HCC recurrence
| AFP model* | |
|---|---|
| Variables | Points |
| Largest tumour diameter (cm) | |
| ≤ 3 | 0 |
| 3-6 | 1 |
| > 6 | 4 |
| Number of nodules | |
| 1-3 | 0 |
| ≥ 4 | 2 |
| AFP level (ng/mL) | |
| ≤ 100 | 0 |
| 100-1000 | 2 |
| > 1000 | 3 |
The AFP score was subsequently validated in a cohort of 574 patients with a high prevalence of viral hepatitis as an aetiologic factor for chronic liver disease, who were transplanted for HCC in 4 Italian centres[26]. An AFP score ≤ 2 again identified a group of patients with low risk of recurrence, even if they were beyond the Milan criteria at listing. Additionally, in a subgroup of patients who underwent a downstaging procedure prior to listing, an AFP score ≤ 2 was associated with an excellent 5 year survival and reduced risk of recurrence[26]. Another validation study which included 327 patients from Latin America has also demonstrated the superiority of the AFP score compared to Milan criteria in predicting post LT recurrence, even in patients who were downstaged in order to fulfil the listing criteria[27].
In a study that aimed to examine the survival benefit and cost-utility in order to better allocate medical resources, the AFP score was proven to be a useful tool for cost-effectiveness[28]. LT was a cost-effective treatment in patients with AFP score ≤ 3 and was proven to be cost-ineffective in patients with AFP score > 7. Although cost-effectiveness should not directly determine eligibility for transplantation, it should be taken into consideration in order to improve organ allocation. Finally, as previously mentioned, in 2013 the AFP model was adopted by the French Organisation for Organ Sharing as the official national listing criteria.
Metroticket 2.0
In a study by Mazzaferro et al.[7] which included in the training set 1018 patients who underwent LT in 3 different centres in Italy, and in the validation set, 341 patients transplanted for HCC in China, a model that consists of the sum of tumour size and number preoperatively and log10AFP, has shown better predictability of recurrence and survival compared to Milan criteria. By using three different cut-off AFP values the authors defined the AFP-adjusted-to-HCC size (AFP-UTS) criteria as shown in Table 3. Patients within compared to beyond the AFP-UTS criteria showed a 5-year overall, HCC-specific and recurrence-free survival of 79.7% vs. 51.2% (P < 0.0001), 93.5% vs. 55.6% (P < 0.0001), and 89.6% vs. 46.8% (P < 0.0001), respectively. An online calculator was also developed (www.hcc-olt-metroticket.org) which provides a 5-year post-transplantation prediction of HCC specific survival based on the pre-operative radiological tumour assessment and the last AFP value. The prediction value can also be refined based on the presence or not of HCV infection, as this can have a negative impact on overall post-transplant survival.
The proposed AFP-UTS criteria for the prediction of HCC recurrence
| AFP-UTS criteria |
|---|
| HCC at pre-transplantation radiology within up to 7 criteria*, if AFP < 200 ng/mL |
| HCC at pre-transplantation radiology within up to 5 criteria*, if AFP 200-400 ng/mL |
| HCC at pre-transplantation radiology within up to 4 criteria*, if AFP 400-1000 ng/mL |
RETREAT score
The Risk Estimation of Tumour Recurrence after Transplant (RETREAT) score which consists of the sum of the largest viable tumour diameter and number of viable tumours on explant, microvascular invasion and AFP at the time of LT, was developed in a cohort of 721 patients across 3 U.S centres and externally validated in a cohort of 340 patients from a single centre in Canada. The RETREAT score is calculated as shown in Table 4. Patients with a RETREAT score of 0 have a predicting 1 and 5 year recurrence risk of 1% (95% CI : 0.0%-2.1%) and 2.9% (95% CI : 0.0%-5.6%) respectively which can increase to 29.3% (95% CI : 25.5%-50.5%) and 75.2% (95% CI : 56.7%-85.8%) in patients with a RETREAT score of 5 or higher. One of the advantages of the RETREAT score over other proposed scoring systems is that it takes into consideration the effect of pre-transplant locoregional treatment by including only viable tumours into the model equation. Although this score can only be calculated post LT, it can be utilised to determine surveillance strategies, as well as influence decisions on immunosuppression regimens and adjuvant therapies post LT.
The RETREAT score for the prediction of HCC recurrence
| RETREAT score | |
|---|---|
| Predictor | Retreat points |
| AFP at LT, ng/mL | |
| 0-20 | 0 |
| 21-99 | 1 |
| 100-999 | 2 |
| ≥ 1000 | 3 |
| Microvascular invasion | 2 |
| Largest viable tumour diameter (cm) plus No of viable tumours* | |
| 0 | 0 |
| 1.1-4.9 | 1 |
| 5-9.9 | 2 |
| ≥ 10 | 3 |
In a study by Mehta et al.[13], the RETREAT score was validated by using the United Network for Organ Sharing database in 3275 patients transplanted for HCC, between 2012 and 2014. Based on explant findings, the RETREAT score discriminated well between patients with low and high risk and recurrence and higher scores were associated with poor survival outcomes. Specifically, patients with a RETREAT score of 0 had a 3 year recurrence and survival rate of 1.6% and 91% respectively, whereas, for patients with a RETREAT score of 5 or higher, the 3 year recurrence and survival rates were 29% and 58% accordingly. The RETREAT score was also shown to be superior to Milan criteria on explant, in predicting HCC recurrence. Finally, the RETREAT score was associated with a shorter time to HCC recurrence with a median time to recurrence of 10.9 months (IQR 51, -17.9) in patients with a score ≥ 4[13].
HALT HCC score
The Hazzard associated with Liver Transplantation for Hepatocellular Carcinoma (HALT-HCC) score was developed based on retrospective data from 420 patients transplanted for HCC in a single US centre, and included MELD-sodium (MELD-Na), tumour burden score (tumour maximum diameter plus number of lesions) and AFP as shown in the following equation; HALT-HCC = (1.27 × tumour burden score) + (1.85 × lnAFP) + (0.26 × MELD-Na)[29]. The HALT-HCC score was externally validated in 13,717 patients that derived from the SRTR and was significantly associated with overall survival (HR 1.06%, 95% CI : 1.05-1.07). Patients were shown to have similar risk of death when stratified by the HALT HCC score, regardless of being within or beyond the Milan criteria prior to transplantation. The advantage of the HALT-HCC score over the other published scores is that it takes into consideration not only the tumour burden and the biological behaviour, but also the underlying liver function at the time of LT[29].
TRAIN score
In a study by Lai et al.[30], a prognostic score that included radiological response criteria (mRESIST), AFP slope, neutrophil to lymphocyte ratio (NLR) prior to transplantation and waiting time (WT) on the transplant list was developed following retrospective analysis of a single cohort (n = 179). The Time-radiological response-AFP-Inflammation (TRAIN) score is calculated as shown in the equation; 0.988 (if mRESIST-progressive disease) + 0.838 (if AFP slope ≥ 15 ng/mL/month) + 0.452 (if NLR ≥ 5.0)-0.03*WT9 (x month). A Score ≥ 1 has shown an excellent ability to stratify patient in terms of intention to treat survival and recurrence. The TRAIN score allowed a potential 8.9% increase in the patients eligible for transplantation without increasing the recurrence risk that would have otherwise been excluded based on the Milan criteria. One of the main disadvantages of this proposed score is that it can only be applied in patients who have undergone pre-transplant locoregional treatment.
The prognostic role of AFP in the context of LT as reported in national and international guidelines
Despite the emerging robust evidence on the ability of pre-operative AFP to predict pre-transplant recurrence, only few International transplant societies have implemented this as part of their listing criteria.
The recently revised EASL Clinical Practice guidelines recommend that the conservative Milan criteria are the benchmark for selection of patients with HCC for LT[2]. Despite this, it is suggested that composite criteria that consider surrogates of tumour biology, (among which AFP is the most relevant), in combination with tumour size and number of nodules is likely to replace conventional criteria for defining eligibility for LT. Although, different cut-off levels have been proposed (100 ng/mL, 200 ng/mL, 400 ng/mL, 1000 ng/mL) no consensus has been reached as to the optimal cut-off level that would best predict HCC recurrence.
In the UK, patients are eligible for LT if they have a single tumour ≤ 5 cm diameter or up to 5 tumours all ≤ 3 cm or a single tumour > 5 cm and ≤ 7 cm diameter, where there has been no evidence of tumour progression (volume increase by < 20%), no extra-hepatic spread and no new nodule formation over a 6-month period. Since 2012, a cut-off AFP level more than 1000 i.u/mL has been used as an exclusion criterion for listing. Tumour rupture, extra-hepatic spread and macrovascular invasion are also considered as absolute contraindications for transplantation[31].
In 2013, the French Organization for Organ Sharing officially implemented the AFP model[10] as the national listing criteria for transplantation. This includes AFP at different cut off levels in combination with tumour size and number[10]. A simplified version of the original model has been proposed [Table 2] and has shown to be superior to Milan criteria in predicting HCC recurrence. The AFP model has been externally validated[26] and has been shown to discriminate well between patients with low and high risk of recurrence, both in patients within or beyond Milan criteria.
In 2008, the Canadian Society for Transplantation recommended that programs use a combination of total tumour volume (TTV), and AFP as listing criteria[9]. Patients are eligible for transplantation if they have TTV ≤ 115 cm3 and AFP ≤ 400 ng/mL. Unlike the Milan criteria, the current score allows the eligibility of patients with more than three tumours but with low tumour volume, which has been associated with favourable outcomes[9]. The TTV is calculated as the sum of the volume of each tumour [(4/3)πr3] based on the maximum radius of each tumour.
Finally, the international consensus conference on LT for HCC which was held in Zurich in 2010 and established internationally accepted statements and guidelines for the conduct of LT, suggested that AFP should be utilised for waiting-list monitoring and following bridging therapy to discriminate between patients with low and high risk for drop out[32].
Conclusion
Successful eradication of hepatitis C virus, following the widespread use and efficacy of the direct acting antiviral (DAA) treatment, has already reduced the number of patients with HCV related cirrhosis requiring LT. As a result, HCC and non-alcoholic fatty liver disease are becoming the leading causes for LT in the USA and in Europe. Transplantation for HCC is challenging, as one has to ensure that disease-free survival remains similar to that of patients transplanted due to benign disease. Similarly, it is equally important to ensure that HCC patients are not disadvantaged and erroneously excluded from liver transplantation, based on tumour volume characteristics alone. It is likely therefore that with the reduction of the HCV burden, transplant programs will become less restrictive. Moreover, with alternative biomarkers and the use of liquid biopsies as prognostic tools in HCC, a more “biological” rather than “morphological” approach to HCC treatment is anticipated.
Until these more refined methods for selecting patients become available, existing evidence supports the use of AFP in decision models for LT. Whether an AFP slope can be more informative compared to a static single value remains unclear. Of the currently available models the “AFP model” is currently the most extensively utilised and validated. The Metroticket 2.0 calculator allows an individualised accurate prediction of post LT recurrence and can be used by different transplant programs, which can be more permissive or restrictive based on the recurrence rates they are willing to accept. Finally, models that incorporate AFP at different cut-offs have shown be superior to Milan criteria in predicting recurrence, but require to be prospectively validated in order to confirm their prognostic value.
Declarations
Authors’ contributionsDrafted the manuscript: Fatourou EM
Reviewed the manuscript and made the appropriate corrections: Suddle AR, Heneghan MA
Availability of data and materialsNot applicable.
Financial support and sponsorshipHeneghan MA is funded by research support from the King’s College Hospital Charity (Orpin Bequest and Kelly Group), and the European Association for the Study of Liver.
Conflicts of interestBoth authors declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2018.
REFERENCES
1. Yang JD, Larson JJ, Watt KD, Allen AM, Wiesner RH, Gores GJ, Roberts LR, Heimbach JA, Leise MD. Hepatocellular carcinoma is the most common indication for liver transplantation and placement on the waitlist in the United States. Clin Gastroenterol Hepatol 2017;15:767-75.
2. European Association for the Study of the Liver. Electronic address: easloffice@easloffice.eu; European Association for the Study of the Liver. EASL practice guidelines: management of hepatocellular carcinoma. J Hepatol 2018;69:182-236.
3. Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, Montalto F, Ammatuna M, Morabito A, Gennari L. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 1996;334:693-9.
4. Lerut J, Lai Q. Morphology does not tell us the entire story: biological behavior improves our ability to select patients with hepatocellular carcinoma waiting for liver transplantation. Hepatobiliary Pancreat Dis Int 2015;14:570-1.
5. Lai Q, Iesari S, Melandro F, Mennini G, Rossi M, Lerut J. The growing impact of alpha-fetoprotein in the field of liver transplantation for hepatocellular cancer: time for a revolution. Transl Gastroenterol Hepatol 2017;2:72.
6. Merani S, Majno P, Kneteman NM, Berney T, Morel P, Mentha G, Toso C. The impact of waiting list alpha-fetoprotein changes on the outcome of liver transplant for hepatocellular carcinoma. J Hepatol 2011;55:814-9.
7. Mazzaferro V, Sposito C, Zhou J, Pinna AD, De Carlis L, Fan J, Cescon M, Di Sandro S, Yi-Feng H, Lauterio A, Bongini M, Cucchetti A. Metroticket 2.0 model for analysis of competing risks of death after liver transplantation for hepatocellular carcinoma. Gastroenterology 2018;154:128-39.
8. Mehta N, Heimbach J, Harnois DM, Sapisochin G, Dodge JL, Lee D, Burns JM, Sanchez W, Greig PD, Grant DR, Roberts JP, Yao FY. Validation of a risk estimation of tumor recurrence after transplant (RETREAT) score for hepatocellular carcinoma recurrence after liver transplant. JAMA Oncol 2017;3:493-500.
9. Toso C, Meeberg G, Hernandez-Alejandro R, Dufour JF, Marotta P, Majno P, Kneteman NM. Total tumor volume and alpha-fetoprotein for selection of transplant candidates with hepatocellular carcinoma: a prospective validation. Hepatology 2015;62:158-65.
10. Duvoux C, Roudot-Thoraval F, Decaens T, Pessione F, Badran H, Piardi T, Francoz C, Compagnon P, Vanlemmens C, Dumortier J, Dharancy S, Gugenheim J, Bernard PH, Adam R, Radenne S, Muscari F, Conti F, Hardwigsen J, Pageaux GP, Chazouilleres O, Salame E, Hilleret MN, Lebray P, Abergel A, Debette-Gratien M, Kluger MD, Mallat A, Azoulay D, Cherqui D. Liver transplantation for hepatocellular carcinoma: a model including alpha-fetoprotein improves the performance of Milan criteria. Gastroenterology 2012;143:986-94.
11. Hameed B, Mehta N, Sapisochin G, Roberts JP, Yao FY. Alpha-fetoprotein level > 1000 ng/mL as an exclusion criterion for liver transplantation in patients with hepatocellular carcinoma meeting the Milan criteria. Liver Transpl 2014;20:945-51.
12. Lai Q, Avolio AW, Graziadei I, Otto G, Rossi M, Tisone G, Goffette P, Vogel W, Pitton MB, Lerut J. Alpha-fetoprotein and modified response evaluation criteria in solid tumors progression after locoregional therapy as predictors of hepatocellular cancer recurrence and death after transplantation. Liver Transpl 2013;19:1108-18.
13. Mehta N, Dodge JL, Roberts JP, Yao FY. Validation of the prognostic power of the RETREAT score for hepatocellular carcinoma recurrence using the UNOS database. Am J Transplant 2018;18:1206-13.
14. Hakeem AR, Young RS, Marangoni G, Lodge JP, Prasad KR. Systematic review: the prognostic role of alpha-fetoprotein following liver transplantation for hepatocellular carcinoma. Aliment Pharmacol Ther 2012;35:987-99.
15. Berry K, Ioannou GN. Serum alpha-fetoprotein level independently predicts posttransplant survival in patients with hepatocellular carcinoma. Liver Transpl 2013;19:634-45.
16. Lai Q, Avolio AW, Manzia TM, Sorge R, Agnes S, Tisone G, Berloco PB, Rossi M. Combination of biological and morphological parameters for the selection of patients with hepatocellular carcinoma waiting for liver transplantation. Clin Transplant 2012;26:E125-31.
17. Vibert E, Azoulay D, Hoti E, Iacopinelli S, Samuel D, Salloum C, Lemoine A, Bismuth H, Castaing D, Adam R. Progression of alphafetoprotein before liver transplantation for hepatocellular carcinoma in cirrhotic patients: a critical factor. Am J Transplant 2010;10:129-37.
18. Lai Q, Vitale A, Iesari S, Finkenstedt A, Mennini G, Spoletini G, Hoppe-Lotichius M, Vennarecci G, Manzia TM, Nicolini D, Avolio AW, Frigo AC, Graziadei I, Rossi M, Tsochatzis E, Otto G, Ettorre GM, Tisone G, Vivarelli M, Agnes S, Cillo U, Lerut J. Intention-to-treat survival benefit of liver transplantation in patients with hepatocellular cancer. Hepatology 2017;66:1910-19.
19. Toso C, Asthana S, Bigam DL, Shapiro AM, Kneteman NM. Reassessing selection criteria prior to liver transplantation for hepatocellular carcinoma utilizing the scientific registry of transplant recipients database. Hepatology 2009;49:832-8.
20. Ciccarelli O, Lai Q, Goffette P, Finet P, De Reyck C, Roggen F, Sempoux C, Doffagne E, Reding R, Lerut J. Liver transplantation for hepatocellular cancer: UCL experience in 137 adult cirrhotic patients. Alpha-foetoprotein level and locoregional treatment as refined selection criteria. Transpl Int 2012;25:867-75.
21. Zheng SS, Xu X, Wu J, Chen J, Wang WL, Zhang M, Liang TB, Wu LM. Liver transplantation for hepatocellular carcinoma: Hangzhou experiences. Transplantation 2008;85:1726-32.
22. McHugh PP, Gilbert J, Vera S, Koch A, Ranjan D, Gedaly R. Alpha-fetoprotein and tumour size are associated with microvascular invasion in explanted livers of patients undergoing transplantation with hepatocellular carcinoma. HPB (Oxford) 2010;12:56-61.
23. Grat M, Wronka KM, Stypulkowski J, Bik E, Krasnodebski M, Masior L, Lewandowski Z, Grat K, Patkowski W, Krawczyk M. The Warsaw proposal for the use of extended selection criteria in liver transplantation for hepatocellular cancer. Ann Surg Oncol 2017;24:526-34.
24. Lee HW, Song GW, Lee SG, Kim JM, Joh JW, Han DH, Kim SI, Kim SH, Kim DS, Cho JY, Suh KS. Patient selection by tumor markers in liver transplantation for advanced hepatocellular carcinoma. Liver Transpl ; doi: 10.1002/lt.25056.
25. She WH, Chan ACY, Cheung TT, Lo CM, Chok KSH. Survival outcomes of liver transplantation for hepatocellular carcinoma in patients with normal, high and very high preoperative alpha-fetoprotein levels. World J Hepatol 2018;10:308-18.
26. Notarpaolo A, Layese R, Magistri P, Gambato M, Colledan M, Magini G, Miglioresi L, Vitale A, Vennarecci G, Ambrosio CD, Burra P, Di Benedetto F, Fagiuoli S, Colasanti M, Maria Ettorre G, Andreoli A, Cillo U, Laurent A, Katsahian S, Audureau E, Roudot-Thoraval F, Duvoux C. Validation of the AFP model as a predictor of HCC recurrence in patients with viral hepatitis-related cirrhosis who had received a liver transplant for HCC. J Hepatol 2017;66:552-59.
27. Pinero F, Tisi Bana M, de Ataide EC, Hoyos Duque S, Marciano S, Varon A, Anders M, Zerega A, Menendez J, Zapata R, Munoz L, Padilla Machaca M, Soza A, McCormack L, Poniachik J, Podesta LG, Gadano A, Boin IS, Duvoux C, Silva M. Liver transplantation for hepatocellular carcinoma: evaluation of the alpha-fetoprotein model in a multicenter cohort from Latin America. Liver Int 2016;36:1657-67.
28. Vitale A, Farinati F, Burra P, Trevisani F, Giannini EG, Ciccarese F, Piscaglia F, Rapaccini GL, Di Marco M, Caturelli E, Zoli M, Borzio F, Cabibbo G, Felder M, Sacco R, Morisco F, Missale G, Foschi FG, Gasbarrini A, Svegliati Baroni G, Virdone R, Chiaramonte M, Spolverato G, Cillo U. Utility-based criteria for selecting patients with hepatocellular carcinoma for liver transplantation: a multicenter cohort study using the alpha-fetoprotein model as a survival predictor. Liver Transpl 2015;21:1250-8.
29. Sasaki K, Firl DJ, Hashimoto K, Fujiki M, Diago-Uso T, Quintini C, Eghtesad B, Fung JJ, Aucejo FN, Miller CM. Development and validation of the HALT-HCC score to predict mortality in liver transplant recipients with hepatocellular carcinoma: a retrospective cohort analysis. Lancet Gastroenterol Hepatol 2017;2:595-603.
30. Lai Q, Nicolini D, Inostroza Nunez M, Iesari S, Goffette P, Agostini A, Giovagnoni A, Vivarelli M, Lerut J. A novel prognostic index in patients with hepatocellular cancer waiting for liver transplantation: time-radiological-response-alpha-fetoprotein-inflammation (TRAIN) score. Ann Surg 2016;264:787-96.
31. Introduction to patient selection and organ allocation policies. Available from: https://nhsbtdbe.blob.core.windows.net/umbraco-assets-corp/4955/introduction_to_selection_and_allocation_policies.pdf. [Last accessed on 30 Aug 2018].
32. Clavien PA, Lesurtel M, Bossuyt PM, Gores GJ, Langer B, Perrier A. Recommendations for liver transplantation for hepatocellular carcinoma: an international consensus conference report. Lancet Oncol 2012;13:e11-22.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Fatourou EM, Suddle AR, Heneghan MA. Alpha-fetoprotein as a predictor of hepatocellular carcinoma recurrence following liver transplantation. Hepatoma Res 2018;4:63. http://dx.doi.org/10.20517/2394-5079.2018.62
AMA Style
Fatourou EM, Suddle AR, Heneghan MA. Alpha-fetoprotein as a predictor of hepatocellular carcinoma recurrence following liver transplantation. Hepatoma Research. 2018; 4: 63. http://dx.doi.org/10.20517/2394-5079.2018.62
Chicago/Turabian Style
Fatourou, Evangelia M., Abid R. Suddle, Michael A. Heneghan. 2018. "Alpha-fetoprotein as a predictor of hepatocellular carcinoma recurrence following liver transplantation" Hepatoma Research. 4: 63. http://dx.doi.org/10.20517/2394-5079.2018.62
ACS Style
Fatourou, EM.; Suddle AR.; Heneghan MA. Alpha-fetoprotein as a predictor of hepatocellular carcinoma recurrence following liver transplantation. Hepatoma. Res. 2018, 4, 63. http://dx.doi.org/10.20517/2394-5079.2018.62
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 5 clicks
Cite This Article 5 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.