Association of serum levels of transforming growth factor β1 with disease severity in patients with hepatocellular carcinoma
Abstract
Aim: Transforming growth factor (TGF) is overexpressed by tumor cells like other proteins and growth factors. TGF-β1 is then activated in the extracellular compartment but is unable to control cell proliferation because of the absence or low level of TGF-β1 receptors on the plasma membrane of malignant hepatocytes. This potential mechanism might interrupt the autocrine regulation loop of TGF-β1 and its blocking effect on cell proliferation. TGF-β1 is a multifunctional cytokine involved in the regulation of growth and differentiation of both normal and transformed cells. This study aimed to evaluate the association of serum levels of TGF-β1 with disease severity.
Methods: A total of 180 subjects were classified into 6 groups according to Barcelona clinic liver cancer (BCLC) classification, 30 patients each: early (BCLC 0 and A), intermediate (BCLC B), advanced stage (BCLC C), and terminal stage (BCLC D) of hepatocellular carcinoma as well as 1 group of patients with cirrhosis only and 1 control group. Serum levels of TGF-β1 were measured.
Results: Serum levels of TGF-β1 were significantly higher in patients with HCC (1,687.47 ± 1,462.81 pg/mL) than cirrhotics (487.98 ± 344.23 pg/mL, P < 0.001) and controls (250.16 ± 284.61 pg/mL, P < 0.001).
Conclusion: TGF-β1 may have a role in tumor growth and progression.
Keywords
Introduction
Liver cancer is the sixth most common cancer, the third cause of cancer related deaths, and accounts for 7% of all cancers[1]. The main risk factors for hepatocellular carcinoma (HCC) are chronic infections with either hepatitis B virus (HBV) or hepatitis C virus (HCV), making up approximately 75%-85% of all cases, as well as excessive alcohol consumption, which is responsible for about 40% of HCC development in Western countries[2]. Chronic inflammation and tissue damage by these agents leads to cirrhosis which is the underlying condition for the majority of HCC cases[3]. Early detection and appropriate treatment remain the best strategy for reducing mortality.
Transforming growth factor β (TGF-β) superfamily is known to be involved in embryonic development, adult tissue homeostasis, and disease pathogenesis. Specifically, it has been shown to control proliferation, differentiation, apoptosis, migration, extracellular matrix remodeling, immune functions, and tumor invasion/metastasis[4]. TGF-β enhances hepatic stellate cell activation, stimulates collagen gene transcription and suppresses matrix metalloproteinases expression. Thus, TGF-β, as well as its intracellular mediators; Smad proteins, can be potential therapeutic targets for liver fibrosis. TGF-β inhibits hepatocyte proliferation, but it also promotes HCC. TGF-β has been shown to play both tumor-suppressive at early stage and tumor-promoting roles at later stage[5]. At the early stage of tumorogenesis, TGF-β1 inhibited normal cell growth and tumorogenesis by suppressing G1/S phase transition[6], in later stages; malignant cells become resistant to suppressive effects of TGF-β either through mutation and/or functional inactivation of TGF-β receptors or by downstream alterations in the SMAD-signaling pathway[7]. Mutations in downstream TGF-β signaling components cause variable attenuations or complete loss of expression; these mutations, which have been detected in many common tumors, affect TGF-β signal transmission that potentially results in human cancer development and progression[8]. TGF-β1 expression was related to tumor grade and pathological stage. Furthermore, overexpression of plasma TGF-β1 was associated with invasiveness of HCC and worse prognosis[9].
The aim of this study was to evaluate the association between serum level of TGF-β1 and disease severity in Egyptian patients with HCC.
Methods
This cross sectional study was conducted at National Liver Institute, Menoufia University. The study protocol was approved by institute Ethics Committee. A written informed consent was obtained from all participants in the study.
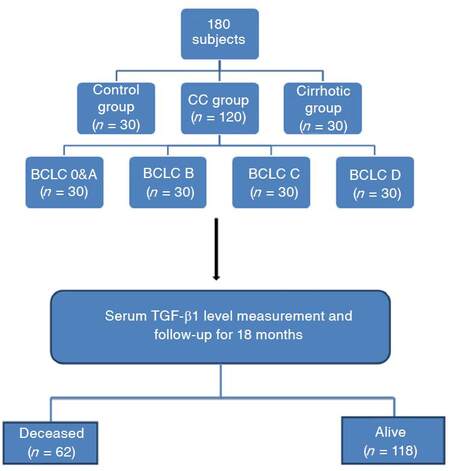
The study was performed on 180 subjects attending HCC and cirrhosis clinics, 120 HCC patients, 30 cirrhotic patients and 30 matched apparently healthy subjects served as control group. HCC patients were classified according to Barcelona clinic liver cancer (BCLC) classification into 6 groups: group 1 comprised 30 patients with an early HCC stage (BCLC 0 and A); group 2 comprised 30 patients with HCC intermediate stage (BCLC B); group 3 comprised 30 patients with an advanced HCC stage (BCLC C); group 4 comprised 30 patients with a terminal HCC stage (BCLC D); group 5 comprised 30 patients with cirrhosis without evidence of HCC; and group 6 comprised 30 healthy subjects as a control group [Figure 1].
Figure 1. Flow chart of the study. BCLC: Barcelona clinic liver cancer; TGF-β1: transforming growth factor beta 1; HCC: hepatocellular carcinoma
The diagnosis of HCC was based on non-invasive criteria using multi-slice triphasic spiral computed tomography or contrast enhanced dynamic magnetic resonance imaging. The presence of typical features of arterial enhancement and rapid portal or delayed washout on one imaging technique was diagnostic of HCC for nodules > 2 cm in diameter in cirrhotic patients. In cases of uncertainty or atypical radiological findings, diagnosis was confirmed by biopsy[10]. Liver cirrhosis was diagnosed by ultrasonographical findings (shrunken liver, coarse echo pattern, attenuated hepatic veins and nodular surface) and biochemical indication of parenchymal harm.
Laboratory investigations
Venous blood (10 mL) were drawn from all participants and divided into 3 parts: the 1st part, 2 mL was collected in EDTA containing tube for complete blood picture using Sysmex K-21, (Sysmex Corporation, Kobe, Japan); the 2nd part, 5 mL for serum which was used for assessment of liver function tests using fully automated autoanalyzer SYNCHRON CX9ALX (Beckman Coulter Inc., CA, USA), for immunoassay HBs Ag and HCV Ab (Abbott Laboratories, Abbott Park, IL, USA), and for serum AFP level measurement using the automated chemiluminescence system ACS 180 (Siemens Medical Solutions, USA); and the 3rd part, 3 mL for serum and was used for measurement of TGF-β1 using human TGF-β1 ELISA Kit (New York, NY 10123) according to the manufacturer’s instructions.
Statistical analysis
Data was statistically analyzed using SPSS (Statistical Package for Social Science) Program version 13 for Windows and for all the analysis a P value < 0.05 was considered statistically significant.
Results
The demographic criteria of all subjects included in the study are shown in Table 1.
Demographic criteria in all subjects
| Control | Cirrhotic | HCC | χ2 | P value | ||||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |||
| Gender | ||||||||
| Male | 16 | 53.3 | 20 | 66.7 | 99 | 82.5 | 12.222 | 0.003 |
| Female | 14 | 46.7 | 10 | 33.3 | 21 | 17.5 | ||
| Age, years, mean ± SD | 47.67 ± 3.387 | 52.29 ± 2.997 | 50.20 ± 3.916 | 26.2 | 0.001 | |||
| Residence | ||||||||
| Rural | 15 | 50 | 10 | 33.3 | 95 | 79.1 | 27.19 | 0.001 |
| Urban | 15 | 50 | 20 | 66.6 | 25 | 20.8 | ||
| Occupation | ||||||||
| House wife | 8 | 26.6 | 15 | 50 | 17 | 14.1 | 20.28 | 0.004 |
| Farmer | 15 | 50 | 10 | 33.3 | 55 | 45.8 | ||
| Employee | 7 | 23.3 | 5 | 16.6 | 48 | 40 | ||
| Smoking | ||||||||
| Non | 19 | 63.3 | 20 | 66.7 | 56 | 46.7 | 9.240 | 0.055 |
| EX | 0 | 0 | 4 | 13.3 | 16 | 13.3 | ||
| Yes | 11 | 36.7 | 6 | 20.0 | 48 | 40.0 | ||
| Pesticidal exposure | ||||||||
| No | 15 | 50 | 14 | 46.7 | 65 | 54.2 | 0.612 | 0.736 |
| Yes | 15 | 50 | 16 | 53.3 | 55 | 45.8 | ||
| Bilharziasis or anti bilharzial | ||||||||
| No | 22 | 73.3 | 17 | 56.7 | 23 | 19.2 | 39.058 | 0.001 |
| Yes | 8 | 26.7 | 13 | 43.3 | 97 | 80.8 | ||
| Antiviral treatment | ||||||||
| No | 30 | 100 | 22 | 73.3 | 109 | 90.8 | ||
| Yes | 0 | 0 | 8 | 26.7 | 11 | 9.2 | 12.033 | 0.002 |
| DM | ||||||||
| No | 22 | 73.3 | 22 | 73.3 | 91 | 75.8 | 0.133 | 0.936 |
| Yes | 8 | 26.7 | 8 | 26.7 | 29 | 24.2 | ||
| BCLC | NA | NA | NA | NA | NA | NA | ||
| A | 30 | 25.0 | ||||||
| B | 30 | 25.0 | ||||||
| C | 30 | 25.0 | ||||||
| D | 30 | 25.0 | ||||||
| Child | ||||||||
| A | NA | NA | 29 | 96.7 | 50 | 41.7 | 29.1 | 0.001 |
| B | 1 | 3.3 | 40 | 33.3 | ||||
| C | 0 | 0.0 | 30 | 25.0 | ||||
Table 2 shows the tumor characteristics in 120 patients with HCC included in the study.
Tumor characteristics in HCC patients
| Variable | Number | Percent (%) | |
|---|---|---|---|
| Site of nodule(s) | |||
| Bi-lobar | 54 | 45 | |
| Uni-lobar | 66 | 55 | |
| Right lobe | 48 | 40 | |
| Left lobe | 18 | 15 | |
| Number of nodules | |||
| Single | 58 | 48.3 | |
| Multiple | 62 | 51.6 | |
| 2 | 18 | 15 | |
| 3 | 6 | 5 | |
| > 3 | 38 | 31.6 | |
| Size of nodule(s) | |||
| Largest diameter for single or sum of largest diameter (2-3 nodules) | < 5 cm | 47 | 39.1 |
| 5-8 cm | 18 | 15 | |
| > 8 cm | 17 | 14.1 | |
| Size of largest (> 3 nodules) | 5-8 cm | 6 | 5 |
| > 8 cm | 32 | 26.6 | |
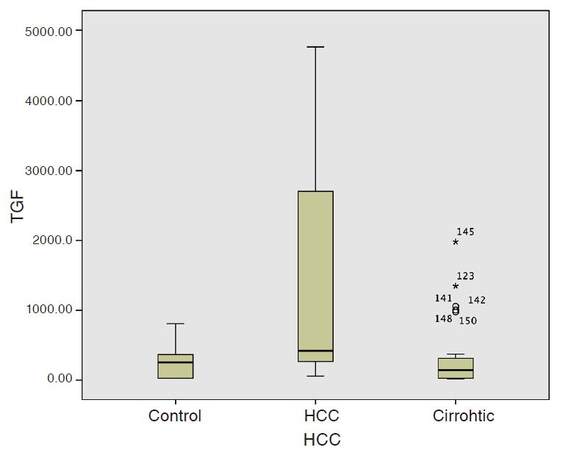
Serum levels of TGF-β1 was significantly higher in HCC groups compared to cirrhotic and control groups (P = 0.000) as shown in Table 3 and Figure 2.
Serum levels of TGF-β1 in all subjects
| Variables | The studied groups (mean ± SD) | Kruskal Wallis test | P-value | ||
|---|---|---|---|---|---|
| HCC (n = 120) | Cirrhotic (n = 30) | Control (n = 30) | |||
| TGF | 1,687.47 ± 1,462.81 | 487.98 ± 344.23 | 250.16 ± 284.61 | 33.990 | 0.000 |
Figure 2. TGF level among the studied groups. TGF-β1: transforming growth factor beta 1; HCC: hepatocellular carcinoma
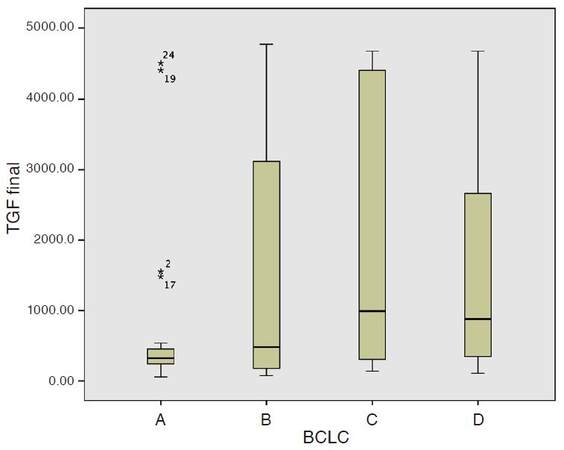
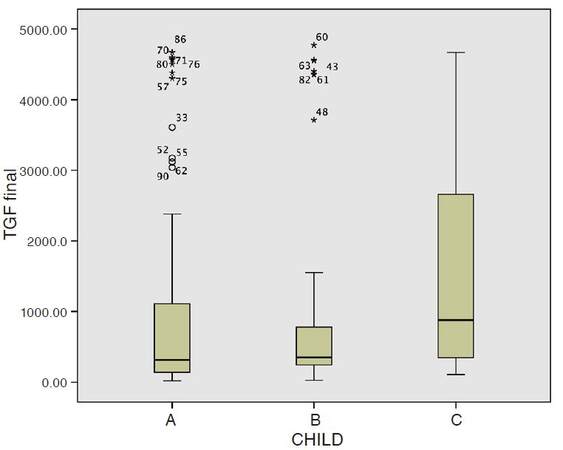
More advanced BCLC stage was generally associated with higher serum levels of TGF-β1, as shown in Table 4 and Figure 3, patients at an early stage HCC (BCLC stage A), had significantly lower serum levels of TGF-β1 compared to BCLC stage C and D (P = 0.004 and 0.038 respectively) but not to BCLC stage B (P = 0.267). Similarly, serum levels of TGF-β1 were significantly higher with more advanced liver disease assessed by Child Pugh classification [Table 4 and Figure 4].
Serum levels of TGF-β1 according to BCLC and Child-Pugh score
| Variable | TGF (mean ± SD) | Kruskal Wallis test | P value |
|---|---|---|---|
| BCLC stage | |||
| A | 652.83 ± 1084.60 | 12.100 | 0.007 |
| B | 1378.95 ± 1660.50 | ||
| C | 2150.68 ± 1970.01 | ||
| D | 1668.78 ± 1628.15 | ||
| Child classification | |||
| A | 1079.45 ± 1491.016 | 6.729 | 0.035 |
| B | 1232.30 ± 1717.276 | ||
| C | 1668.78 ± 1628.15 | ||
Figure 3. Serum TGF level according to BCLC classification. BCLC: Barcelona clinic liver cancer; TGF-β1: transforming growth factor beta 1
Table 5 shows that higher serum levels of TGF-β1 were significantly associated with vascular and invasion and tumor size (P = 0.001 and 0.02 respectively) rather than number of nodules (P = 0.964).
Serum levels of TGF-β1 and tumor burden (vascular invasion, tumor size and number)
| Tumor burden | TGF (mean ± SD) | Statistical test | P-value |
|---|---|---|---|
| Vascular invasion | Mann Whitney test 3.32 | 0.001 | |
| Yes | 1909.29 ±1872.17 | ||
| No | 1019.65 ±1425.38 | ||
| Tumor size | Kruskal Wallis Test 7.2 | 0.02 | |
| < 5 cm | 1106.06 ± 1541.75 | ||
| 5-8 cm | 1217.14 ± 1522.95 | ||
| > 8 | 1925.32 ± 1815.78 | ||
| Tumor Number | Mann Whitney test 0.002 | 0.964 | |
| Single | 1427.55 ± 1669.07 | ||
| Multiple | 1495.79 ± 1717.45 |
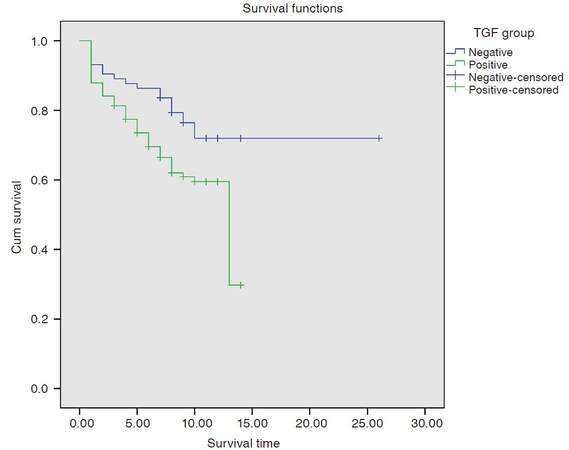
Lower serum levels of TGF-β1 was associated with a higher probability of survival, using a cut-off value of 301.9 pg/mL, the median survival of patients with levels < 301.9 pg/mL was not reached with a probability of survival of 71.9%, the median survival for patients with level ≥ 301.9 pg/mL was only 13 months, the difference was statistically significant using log rank test (P = 0.04), as shown in Tables 6 and 7 and Figure 5.
Number of cases
| TGF | Total number | No. of events | Censored | |
|---|---|---|---|---|
| n | Percent | |||
| Negative < 301 | 73 | 20 | 53 | 72.6% |
| Positive ≥ 301 | 107 | 42 | 65 | 60.7% |
| Overall | 180 | 62 | 118 | 65.6% |
Means and medians for survival time
| TGF score | Mean | Median | ||||||
|---|---|---|---|---|---|---|---|---|
| Estimate | Std. Error | 95% CI | Estimate | Std. Error Lower bound | 95% CI | |||
| Lower bound | Upper bound | Upper bound | ||||||
| Negative < 301 | 11.599 | 0.501 | 10.617 | 12.582 | - | - | - | - |
| Positive > 301 | 9.715 | 0.527 | 8.681 | 10.748 | 13.000 | 2.136 | 8.813 | 17.18 |
| Overall | 10.498 | 0.385 | 9.743 | 11.252 | 13.000 | - | - | - |
Analysis of the receiver operating characteristic (ROC) curve of TGF-β1 showed that, at cut-off value 301.9 pg/mL of TGF-β1; area under the curve for the prediction of HCC was 0.765 and 95%CI 0.694-0.885, with a sensitivity of 72% and a specificity of 65%.
Analysis of the ROC curve of alpha-fetoprotein (AFP) showed that, at cut off value 20 µg/L area of AFP; area under the curve for the prediction of HCC was 0.86 with 95%CI 0.815-0.930 at this cut-off; the sensitivity was 72%, while the specificity was 43%.
Combining both TGF-β1 (at a cut-off value of 301.9 pg/mL) and AFP (at a cut-off value of 20 µg/L) would raise the sensitivity to 90%, but decreasing the specificity to 32%.
Discussion
TGF-β1 acts as a growth inhibitor in normal cells, whereas in tumor cells, it loses the ability to mediate growth inhibition and instead promotes tumor progression by enhancing migration, invasion, and survival of tumor cells[11].
In liver diseases, the persistence of chronic inflammation, as observed in chronic viral hepatitis, plays a major role in determining the shift in the TGF-β1 signaling pathway from tumor suppression to increasing the risk of HCC[12].
Our study evaluated the serum levels of TGF-β1 in HCC patients, cirrhotic patient and normal subjects. Its aim was to determine the specific contribution of TGF-β1 over-expression to progression of HCC.
In our study, we demonstrated significantly higher levels of TGF-β1 in HCC patients (1,687.47 ± 1,462.81 pg/mL) compared to the other two groups (cirrhotic 487.98 ± 344.23 pg/mL and healthy control 250.16 ± 284.61 pg/mL), with no significant difference between control and cirrhotic groups. These findings signified the role of TGF-β1 in tumor growth and progression, implicating its potential use as novel marker for risk prediction of HCC development in cirrhotic patients.
These results are in agreement with Shehata et al.[13], who found that TGF-β1 levels were also significantly higher in patients with HCC group compared to chronic hepatitis C patients and control groups.
Similarly, Lee et al.[9] found that plasma TGF-β1 levels were significantly higher in patients with HCC than in cirrhotic patients and normal controls. However, serum levels of TGF-β1 in the cirrhotic patients were significantly lower than those in normal controls and explained that by decreased synthetic function in patients with advanced cirrhosis, resulting in a lower production of TGF-β1 from hepatocytes themselves.
Our study showed that serum levels of TGF-β1 in HCC patients were associated with more advanced BCLC stages. These findings signified the role of TGF-β1 in tumor growth and progression, implicating its utility as a potential novel marker for risk prediction of HCC progression.
These results are in agreement with Shehata et al.[13], who found that there was a significant difference regarding TGF-β1 between early stage (421.9 ± 105.5 pg/mL) and late stage (769.9 ± 115.8 pg/mL) of HCC patients (P = 0.001).
In our study, serum levels of TGF-β1 were significantly higher in patients with larger tumors, Moreover, higher serum levels of TGF-β1 were associated with vascular invasion, the mean value for tumors without vascular invasion was (1,019.65 ± 1,425.38), while the mean value for tumors with vascular invasion was (1,909.29 ± 1,872.17) (P = 0.001).
Similar results were shown by Lee et al.[9], who found that there was a positive correlation between plasma TGF-β1 concentration and tumor size. These findings suggest that plasma TGF-β1 concentration increases with the invasiveness of HCC making it a novel biomarker for risk prediction of HCC progression.
As cancer develops, cancer cells become more resistant to the growth inhibitory properties of TGF-β1 and both the cancer cells and the stromal cells often increase the production of TGF-β1 which stimulates angiogenesis and cell motility. Also, it suppresses immune response with the extracellular matrix and increases the interaction of tumor cell leading to greater invasiveness and metastatic potential of the cancer[14] acting as a promoter of malignancy during tumor progression[15].
In this study, the serum levels of TGF-β1 was significantly higher in patients with more advanced liver disease, being highest in patients with decompensated Child C cirrhosis and lowest in patients with compensated Child A cirrhosis.
These findings are in agreement with the results of Hussein et al.[16] and Flisiak and Prokopovicz[17], who reported that plasma TGF-β1 was elevated in patients with a higher Child score and also stated that elevated plasma TGF-β1 levels in patients with chronic liver disease might be caused by decreased clearance.
But these results disagreed with Mayoral et al.[18] and Lee et al.[9], who found that the TGF-β1 values decrease significantly with progression of liver dysfunction as assessed by Child-Pugh Score.
The follow-up of our HCC patients for 18 months revealed that: the overall mortality was 51.6% with a median survival of 9 months.
In comparison of the survival rate with plasma TGF-β1 levels, patients with a higher plasma TGF-β1 level (≥ 301 pg/mL) showed significantly lower survival rates than those with a lower plasma TGF-β1 level (< 301 pg/mL) (higher group vs. lower group, 29.8% vs. 71.9% at 18 months). This result in agreement with the result of Lee et al.[9], who found that patients with a higher plasma TGF-β1 levels showed significantly lower survival rates than those with a lower plasma TGF-β1 level ( higher group vs. lower group 47% vs. 60% at 12 months).
The association between high TGF-β1 levels and poor treatment outcomes in advanced HCC patients was anticipated because activation of the TGF-β pathway was linked to angiogenesis and the progression, invasion, and metastasis of cancer cells in late stage malignancies[19].
In this study, ROC curve analysis of TGF-β1 in HCC showed that the best cut-off value of TGF-β1 for detection of HCC patients was 301.9 pg/mL with area under the curve of 0.765 and 95%CI 0.694-0.885, the sensitivity and specificity were 72% and 65% respectively. These results were found to be slightly different from the published reports by Shehata et al.[13], who reported that with a cut off value of TGF-β1 (370 pg/mL); the sensitivity and specificity for differentiation of HCC patients were 86.7% and 100% respectively, whereas area under the curve was found to be 0.97. This difference is mostly due to differences in the study population.
In a systematic review of literature, when AFP of ≥ 20 mcg/L is used as a cut off, the sensitivity of detecting early HCC is reported to be 25%-65% and specificity to be 80%-94%[20]. But here in this study at cut-off point of 20 mcg/L the sensitivity was 72%, while specificity was 43%.
Aiming to increase the sensitivity for early detection of HCC, combination of TGF-β1 and AFP will raise sensitivity to 90% but decreasing specificity to 32%. So TGF-β1 could be complementary to AFP in the diagnosis of HCC, particularly for the cases at an early stage.
Declarations
Authors’ contributionsConcept and design: M.A.S. Kohla, H. Taha
Data acquisition: M.A.S. Kohla, A. Attia, N. Darwesh
Laboratory work: M. Obada
Data analysis: M.A.S. Kohla, A. Attia, N. Darwesh, M. Obada, H. Taha, M.F. Youssef
Statistical analysis: M.F. Youssef
Literature search and manuscript preparation: M.A.S. Kohla, A. Attia, N. Darwesh
Manuscript editing: M.A.S. Kohla, A. Attia, N. Darwesh, H. Taha
Manuscript review: M.A.S. Kohla, A. Attia, N. Darwesh, H. Taha
Financial support and sponsorshipThis study was funded by the National Liver Institute, Menoufia University, Egypt.
Conflicts of interestThere are no conflicts of interest.
Patient consentA written informed consent was obtained from all participants in the study.
Ethics approvalThe study protocol was approved by institute Ethics Committee.
Copyright© The Author(s) 2017.
REFERENCES
1. IRAC, Intermediate Agency for Research on Cancer. Epidemiology of HCC. Available from: https://www.iarc.fr [Last accessed on 1 Jan 2016].
2. Arzumanyan A, Reis HM, Feitelson MA. Pathogenic mechanisms in HBV- and HCV-associated hepatocellular carcinoma. Nat Rev Cancer 2013;13:123-35.
3. Perz JF, Armstrong GL, Farrington LA, Hutin YJF, Bell BP. The contributions of hepatitis B virus and hepatitis C virus infections to cirrhosis and primary liver cancer worldwide. J Hepatol 2006;45:529-38.
4. Guo X, Wang X. Signaling cross-talk between TGF-beta/BMP and other pathways. Cell Res 2009;19:71-88.
5. Bellam N, Pasche B. TGF-β signaling alterations and colon cancer. Cancer Treat Res 2010;155:85-103.
6. Sato Y, Araki H, Kato J, Nakamura K, Kawano Y, Kobune M, Sato T, Miyanishi K, Takayama T, Takahashi M, Takimoto R, Iyama S, Matsunaga T, Ohtani S, Matsuura A, Hamada H, Niitsu Y. Human mesenchymal stem cells xenografted directly to rat liver are differentiated into human hepatocytes without fusion. Blood 2005;106:756-63.
7. Teicher BA. Malignant cells, directors of malignant process: role of transforming growth factor-beta. Cancer Metastasis Rev 2001;20:133-43.
8. Mu-oz-Antonia T, Torrellas-Ruiz M, Clavell J, Mathews LA, Muro-Cacho CA, Bàez A. Aberrant methylation inactivates transforming growth factor beta receptor 1 in head and neck squamous cell carcinoma. Int J Otolrayngol 2009;2009:848695.
9. Lee D, Chung YH, Kim JA, Lee YS, Lee D, Jang MK, Kim KM, Lim YS, Lee HC, Lee YS. Transforming growth factor beta 1 over expression is closely related to invasiveness of hepatocellular carcinoma. Oncology 2012;82:11-8.
10. Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology 2011;53:1020-2.
11. Oh S, Kim E, Kang D, Kim M, Kim JH, Song JJ. Transforming growth factor-beta gene silencing using adenovirus expressing TGF-β1 or TGF-β2 shRNA. Cancer Gene Ther 2013;20:94-100.
12. Lu H, Ouyang W, Huang C. Inflammation, a key event in cancer development. Mol Cancer Res 2006;4:221-33.
13. Shehata F, Abdel Monem N, Sakr M, Kasem S, Balbaa M. Epidermal growth factor, its receptor and transforming growth factor beta 1 in the diagnosis of HCV-induced hepatocellular carcinoma. Med Oncol 2013;30:673.
14. Pérez-Gómez E, Castillo G, Santibanez JF, López-Novoa JM, Bernabéu C, Quintanella M. The role of the TGF-β coreceptor endoglin in cancer. Sci World J 2010;10:2367-84.
15. Shariat SF, Kim JH, Andrew B, Kattan MW, Wheeler TM, Kim IY, Lerner SP, Slawin KM. Preoperative plasma level of transforming growth factor b strongly predict patients outcome in bladder carcinoma. Cancer 2001;92:2985-92.
16. Hussein M, Ibrahim AA, Khattab NF, El-Fouly RF, El-Fouly NF. Serum transforming growth factor beta 1 in hepatitis C virus related chronic liver disease and hepatocellular carcinoma patients. Med J Cairo Uni 2010;78:279-86.
17. Flisiak R, Prokopovicz D. Transforming growth factor beta 1 as a surrogate marker of hepatic dysfunction in chronic liver disease. Clin Chem Lab Med 2000;38:1129-31.
18. Mayoral R, Valverde MA, Izquirdo LC, Gonzàles-Rodríguez A, Boscà L, Martín-Sanz P. Impairment of transforming growth factor beta signaling in Caveolin-1 deficient hepatocytes: role in liver regeneration. J Biol Chem 2010;285:3633-42.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Kohla MAS, Attia A, Darwesh N, Obada M, Taha H, Youssef MF. Association of serum levels of transforming growth factor β1 with disease severity in patients with hepatocellular carcinoma. Hepatoma Res 2017;3:294-301. http://dx.doi.org/10.20517/2394-5079.2017.40
AMA Style
Kohla MAS, Attia A, Darwesh N, Obada M, Taha H, Youssef MF. Association of serum levels of transforming growth factor β1 with disease severity in patients with hepatocellular carcinoma. Hepatoma Research. 2017; 3: 294-301. http://dx.doi.org/10.20517/2394-5079.2017.40
Chicago/Turabian Style
Kohla, Mohamed Ahmed Samy, Ahmed Attia, Nehad Darwesh, Manar Obada, Hossam Taha, Marwa F. Youssef. 2017. "Association of serum levels of transforming growth factor β1 with disease severity in patients with hepatocellular carcinoma" Hepatoma Research. 3: 294-301. http://dx.doi.org/10.20517/2394-5079.2017.40
ACS Style
Kohla, MAS.; Attia A.; Darwesh N.; Obada M.; Taha H.; Youssef MF. Association of serum levels of transforming growth factor β1 with disease severity in patients with hepatocellular carcinoma. Hepatoma. Res. 2017, 3, 294-301. http://dx.doi.org/10.20517/2394-5079.2017.40
About This Article
Copyright
Data & Comments
Data
 Cite This Article 9 clicks
Cite This Article 9 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.