Response rates of hepatocellular carcinoma and hepatic colorectal cancer metastases to drug eluting bead regional liver therapy
Abstract
Aim: The purpose of this study was to evaluate and compare how hepatocellular carcinoma (HCC) and colorectal metastases respond to LC Bead chemoembolization using doxorubicin and irinotecan.
Methods: The authors report their experience with doxorubicin and irinotecan eluting beads to treat 13 patients with primary HCC and 25 patients with colorectal metastases over a 1-year period at a single community based oncology practice. Within the colorectal cancer group they compared irinotecan eluting beads to doxorubicin eluting beads.
Results: Nine of the 11 (81.8%) doxorubicin treated HCC patients had either complete response or partial response. All of the HCC lesions showed reduction in size and tumor enhancement and 10/11 (91%) HCC patients were alive at 24 months post treatment. Fisher’s exact test revealed that among the 22 with colorectal metastases for whom follow-up data were available, those 11 who were treated with doxorubicin were significantly more likely to demonstrate complete or partial response compared to the 11 in the irinotecan treated group (P < 0.001).
Conclusion: Overall, HCC and colon metastasis patients clearly demonstrated the effectiveness of drug eluting beads with 91% of the HCC patients alive 24 months after treatment.
Keywords
Introduction
Primary and secondary malignancies of the liver are very common accounting for more than 530,000 new cases per year.[1] Hepatoma and secondary neoplasms of the liver are expected to increase as the incidence of hepatitis C continues to spread throughout the world. Colorectal metastases to the liver and primary malignant hepatic neoplasms have a poor prognosis with dismal survival rates of 31% at 1 year and 26% at 2 years. Surgery is the definitive treatment for isolated lesions while systemic chemotherapy has been the standard treatment for unresectable liver neoplasms.[1-3] Most lesions are not surgically resectable at the time of diagnosis due to their extensive tumor burden. Treatment strategies for unresectable liver cancer are different for hepatocellular carcinoma (HCC), other primary liver tumors, and metastatic liver cancer. For example, transarterial chemoembolization (TACE) may be the standard for HCC but chemotherapy is still the standard of care for colorectal liver metastasis. TACE has been an effective palliative therapy for malignant tumors of the liver for many years.[4-8] TACE has shown improved patient survival rates compared to conservative treatment for various types of malignant liver tumors.[6-9] The palliative nature of transcatheter embolizations has shown improved patient survival and quality of life as compared to placebo and systemic based chemotherapy.[10] TACE is a useful palliative procedure with its ability to simultaneously infuse concentrated dose of chemotherapeutic drug combined with embolization particles.[5-8] This combination produces elevated local chemotherapeutic drug levels along with vascular occlusion of the feeding vessels killing the tumor resulting in reduced systemic toxicity without causing collateral damage to the surrounding liver parenchyma.
LC Bead drug eluting beads [Biocompatibles UK Ltd, Farnham (a BTG group company)] are approved by the Food and Drug Administration for locoregional embolization. Like conventional TACE, drug eluting beads are available for precision transarterial chemoembolization.[11-13] However, they are different in the way they deliver the drug to the tumor. The beads are compressible sulphonate modified polyvinal alcohol hydrogel microspheres.[14] The drug-eluting beads can be loaded with some positively-charged chemotherapeutic agents such as doxorubicin hydrochroride or irinotecan hydrochloride. There is an ion exchange mechanism which creates the active attraction of the drug to the beads. Just like TACE, the beads are delivered to their exact location with fluoroscopic guided transarterial catheters but this time the drug is loaded into the beads.[5,15] The mixture of beads with doxorubicin or irinotecan can be easily loaded in the pharmacy 2 h prior to delivering them to the patient. The 2 h of soaking allows the drug and beads to interact effectively according to the manufacturer.[16] The controlled release of the drug from the drug eluting beads (DEB) demonstrates very little or no post embolization syndrome as compared to conventional TACE procedures. The LC beads maintain a significantly high intratumoral drug concentration in the tumor bed for a 2-week period. This controlled release process may be more effective than conventional TACE. Systemic toxicity is reduced due to a combination of increase late effects and precise arterial deposition of the beads into the tumor as compared to conventional TACE.
LC Bead embolization can utilize both doxorubicin and irinotecan eluting beads for primary hepatomas, colorectal metastasis and a variety of other liver metastases. The purpose of this study is to determine whether this case series could provide insight into whether treatment methods are associated with treatment response.
Methods
Computed tomography positron emission tomography (CT-PET) and/or magnetic resonance imaging (MRI) studies were reviewed prior to all procedures to guide endovascular treatment [Figure 1]. Four board certified interventional radiologists reviewed all pre procedure imaging for each patient and all 4 actually performed the LC Bead chemoembolizations. This was a retrospective study and no ethical approval was obtained for this study. Informed consent was obtained prior to all interventional procedures. All patients with metastatic colorectal metastases or HCC over a period of 1 year were included in this study. All of the colorectal metastasis patients were treated with systemic chemotherapy prior to endovascular intervention. All patients were treated with drug eluting beads during the study. The time frame between completing chemotherapy and initiating the endovascular treatment was 3-6 months. Subsequently, a follow-up CT-PET scan demonstrated progressive liver metastasis not improved on intravenous chemotherapy. As for the HCC patients, once deemed unresectable, they were included in this study. The decision to treat was based on a multidisciplinary approach including the patient’s oncologist, surgical oncologist and interventional radiologist. The treatment pathway was defined by tumor type and then the appropriate chemotherapeutic agent to be used on that type of liver neoplasm. The treatment pathway included pre-procedural imaging, performing the intra-arterial embolization and then the follow-up CT-PET imaging for evaluation of changes in liver mass. Data were collected and patients were followed by their attending oncologist at routine oncology clinic visits. Our staff reviewed the follow-up outpatient images and results were included in the patient’s electronic medical record for comparison.
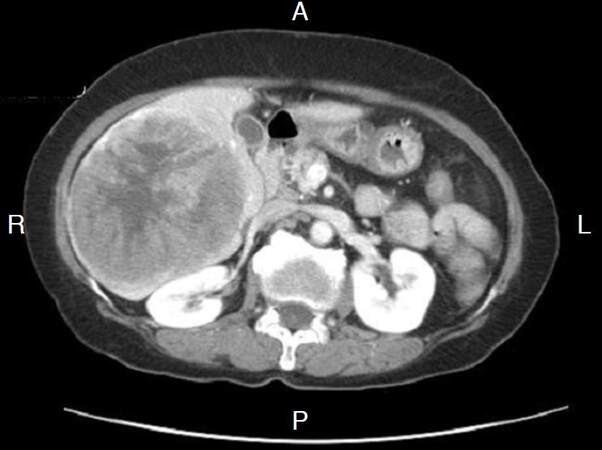
Figure 1. Contrast enhanced computed tomography image of the abdomen demonstrates a large enhancing tumor right hepatic lobe consistent with biopsy proven hepatocellular carcinoma
Pre-procedure images were compared to post treatment images across time to follow response to therapy. Patients were excluded from this study if they had ongoing infection, active gastrointestinal bleeding, liver failure, coagulopathy or allergy to the chemotherapeutic agents. No patients were on Nexavar (Sorafenib) (Bayer HealthCare, Leverkusen, Germany). There was no portal vein invasion in the study patients. No complications due to intra-arterial chemoembolization occurred during the study.
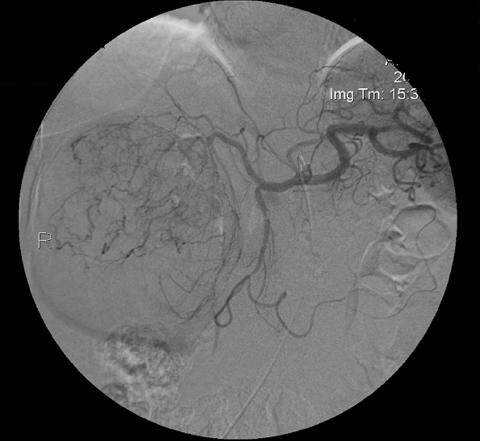
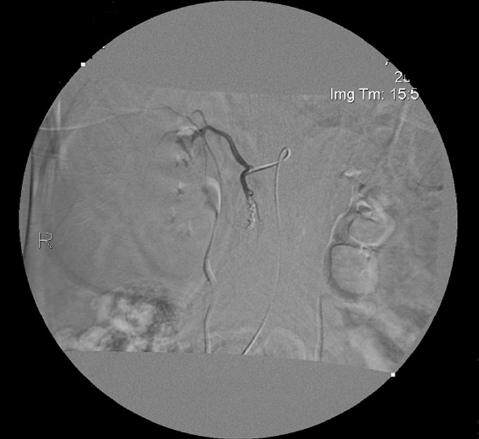
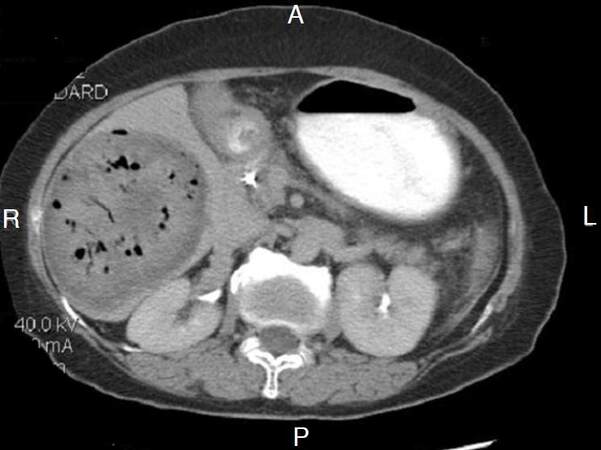
A full angiographic evaluation of all contributing arteries were performed on all patients. A Mariner cobra catheter (Angiodynamics, Latham, NY) was used to perform a pre-embolization angiogram mapping of the hepatic vasculature [Figure 2]. At the discretion of the interventionalist, the gastroduodenal artery was occluded with embolization coils (Target Medical/Boston Scientific Corp. Natick, MA) of various sizes, shapes and number prior to placement of the drug eluting beads. Subsequently, a Renegade microcatheter (Boston Scientific Corp. Natick, MA) was utilized to select various feeding branches during HCC chemoembolization. A more proximal lobar infusion was used for colorectal metastasis chemoembolization due to their more diffuse presentation. The study was performed with 300-500 µm LC Bead which were loaded with either doxorubicin (Bedford Laboratories, Bedford, OH) or irinotecan (Pfizer, Inc., New York, NY) in the hospital pharmacy 2 h prior to the procedure. The doses of irinotecan and doxorubicin were 50 mg/mL and 75 mg/mL respectively and did not change during the study.[1] One 2 mL vial of doxorubicin and irinotecan were mixed with Ominpaque (Iohexol) 350 mg/mL (GE Healthcare Inc, Marlborough, MA) for a total volume of 10 mL. When the beads and drug finished loading, they were deployed through the micro-catheter into the appropriate vascular location. Following deployment of the drug eluted beads, a final angiogram was performed demonstrating no further filling of the neovascular branches to the tumor masses consistent with complete radiographic embolization [Figure 3]. Following the procedure, the patient was monitored overnight for potential discharge the following day. A follow up CT scan was performed the next day to evaluate the embolized tumor [Figure 4]. In 3 months, a follow up PET-CT scan was obtained to evaluate response to the embolization [Figure 5]. In general, all of the pre-treatment images of the HCC patients had similar findings demonstrating significant tumor enhancement on PET-CT. Following LC Bead chemoembolization, there was a significant decrease in size and enhancement of the treated tumor masses exemplified best in Figure 1 and Figure 5. Lesion size, enhancement pattern and metabolic activity were evaluated by the 4 interventionalists on follow-up contrast enhanced CT and/or PET-CT images. Although not included in this manuscript, the HCC patients’ images largely demonstrated heterogeneously enhancing variable sized tumor masses more common in the right hepatic lobe compared to the left. As in the case presented, 2 of the HCC patients had tumors greater than 10 cm in diameter. Most were single lesions without regional adenopathy or metastasis. Two of the patients had multiple liver masses at time of treatment. The TNM staging for the HCC patients in this study ranged from primary tumor T1, T2 and T3a. There were no primary tumors T3b or T4 lesions. There were no regional lymph nodes N0 and no distant metastasis M0.
Figure 3. Post doxorubicin embolization angiogram demonstrates no further visualization of the vascular tumor
Figure 4. Contrast enhanced computed tomography of the abdomen demonstrates gas in tumor the next day post embolization
Figure 5. Three months follow-up positron emission tomography scans demonstrating no uptake within the tumor consistent with complete tumor kill
In total, 36/48 (75%) of the patients received doxorubicin and 12/48 (25%) patients received irinotecan. The majority (52%) of patients had colon metastasis. Of those, 13/25 (52%) received doxorubicin and 12/25 (48%) received irinotecan. Thirteen patients (27%) had HCC and all 13 received doxorubicin. Of the 10 remaining tumor types, all received doxorubicin. Due to changes in practice over the course of the year when patients were treated, the first 12 colon metastasis patients out of the 25 received irinotecan and all the remaining colon metastasis patients received doxorubicin. Initially, the colon metastasis patients received irinotecan based on the chemotherapy data at that time. However, the initial results of the first 12 patients were statistically poor and the investigators replaced irinotecan with doxorubicin on all the remaining patients in the study. Based on the poor results with irinotecan eluting beads for the first 12 colorectal metastasis patients, it was clear that this treatment protocol was not effective and needed to be replaced for the benefit of our patients. Certainly, the results were surprising to our researchers especially according to the results documented in the medical oncology literature related to irinotecan treatment of colorectal metastasis. The medical oncologists reviewed all cases with the interventional radiologists and together agreed to replace the protocol in response to the poor irinotecan results. Furthermore, from the onset of the study, all of the HCC patients received doxorubicin based on the chemotherapy data results at that time.
Frequencies and percentages were used to characterize demographic, clinical, and outcomes data from our consecutive case series. Two outcome categories were created for patients with colorectal metastasis by combining those with partial response or stable disease into one category and those with worsened disease into a second category. Due to the small sample size, a Fisher’s exact test was used to test the hypothesis that among those with colorectal metastases, outcomes (i.e. partial response or stable disease vs. worsened disease) differed according to the type of treatment received (doxorubicin vs. irinotecan). The difference was considered statistically significant at an alpha of 0.05.
Results
A total of 48 patients with unresectable malignant neoplasms of the liver were treated in a 1-year period. There were 28 men (age ranging 34-88 years, with a mean age of 60.5 years) and 20 women (age ranging 34-92 years, with a mean age of 66.2 years). Six patients were lost to follow-up at time of this article. The series includes HCC and colon metastasis [Figure 1]. All of the HCC tumors were hyper-vascular on angiography and became hypo-vascular on follow up scans [Figures 2, 3 and 5]. Many of the remaining tumor types demonstrated hypo-vascular appearance on angiography as compared to HCC. The tumor and treatment types are outlined in Table 1.
Summary of liver neoplasms and treatment type (n = 48)
| Type of neoplasm | Number of patients | Treatment type, n (%) | Number deceased | |
|---|---|---|---|---|
| Doxorubicin | Irinotecan | |||
| Colorectal metastases | 25 | 13 (52) | 12 (48) | 1 |
| Primary hepatoma (HCC) | 13 | 13 (100) | 0 | 1 |
| Breast metastases | 3 | 3 (100) | 0 | |
| Lung metastases | 1 | 1 (100) | 0 | |
| Melanoma metastases | 1 | 1 (100) | 0 | |
| Sarcoma metastases | 1 | 1 (100) | 0 | |
| Pancreatic metastases | 1 | 1 (100) | 0 | |
| Neuroendocrine metastases | 1 | 1 (100) | 0 | |
| Adrenal metastases | 1 | 1 (100) | 0 | |
| Pediatric hepatoblastoma | 1 | 1 (100) | 0 | 1 |
Table 2 shows treatment responses according to tumor and treatment types. Nine of the 11 (81.8%) doxorubicin treated HCC patients had either complete response or partial response. All of the HCC lesions showed reduction in size and tumor enhancement and 10/11 (91%) HCC patients were alive at 24 months post treatment [Table 2]. Fisher’s exact test revealed that among the 22 with colorectal metastases for whom follow-up data were available, those 11 who were treated with doxorubicin were significantly more likely to demonstrate complete or partial response compared to the 11 in the irinotecan treated group (P < 0.001) [Table 3].
Treatment responses for patients according to tumor and treatment types
| Tumor and treatment types | Complete response | Partial response | Worsened | No follow-up |
|---|---|---|---|---|
| Hepatocellular carcinoma | ||||
| Doxorubicin (n = 13) | 8 | 1 | 2* | 2 |
| Colon metastases | ||||
| Doxorubicin (n = 13) | 6 | 4 | 1 | 2 |
| Irinotecan (n = 12) | 0 | 1 | 10* | 1 |
Two-by-two contingency table used to test the hypothesis that among those with colon metastases, those treated with irinotecan had worse outcomes than those treated with doxorubicin
| Complete or partial response | Worsened | |
|---|---|---|
| Doxorubicin treated (n = 11) | 10 | 1 |
| Irinotecan treated (n = 11) | 1 | 10 |
Discussion
Our study compared how HCC and colorectal metastases responded to catheter directed LC Bead emobolization with irinotecan and doxorubicin. The results were compelling for a small sample size. Of the 13 colon cancer study patients who were treated with doxorubicin, 46.2% had a complete response and 4/13 (30.8%) had stable disease. The HCC patients on the other hand improved significantly with 81% demonstrating complete or partial response and 91% of them alive at 24 months after treatment.
Overall, the results of this study demonstrated that many patients with unresectable colon metastasis or HCC who were treated with doxorubicin drug eluting beads demonstrated a complete or partial response. All of these patients treated with doxorubicin who showed complete or partial response remained in remission from liver disease for at least 24 months. However, those colon metastasis patients treated with irinotecan eluting beads did poorly and the study investigators stopped using irinotecan on the remaining patient cohort. Only 1 patient out of 12 (8.3%) demonstrated partial response with irintoecan. Even those patients who responded to systemic irinotecan therapy prior to endovascular treatment did poorly. The irinotecan treated colorectal metastasis patients had poor response rate at 3 months with no reduction in tumor size or tumor enhancement compared to pre-procedural images. The 3 months interval time frame was long enough to account for the post treatment inflammation and edema caused by chemoembolization on the hepatic tumors. Doxorubicin and irinotecan were selected due to the chemotherapy data at that time.
Fiorentini et al.[17] described an 80% response rate following drug eluting bead embolization using irinotecan. However, they used twice the dose of inrinotecan (100 mg/mL) compared to this study. Furthermore, their patients were treated once every 3 weeks and subsequently demonstrated improvement in contrast enhancement on all responding patients. In comparison, this article used the standard dosage which may not have been concentrated enough and/or the treatment time may not have been long enough for the embolization to obtain this type of response. Also in their study, the embolization treatments were stopped if findings of progressive disease were noted and subsequently those patients were excluded from the study. On the other hand, our study included all the patients treated with one session of irinotecan bead embolization and none were excluded from the study despite the results.
Along with chemoembolization, combination therapies including radiofrequency ablation, microwave ablation and cryoablation can be used in conjunction with synergistic effects.[18] The idea of combination therapies is to both embolize the larger tumors decreasing the size with the DEB and then percutaneously ablate the remaining tumor. The DEB treatment prior to percutaneous ablation devascularizes the surface of the tumor which reduces the heat-sink making ablation more effective. Percutaneous ablation of the center of the tumor mass results in a sub lethal temperature experienced at the periphery of the tumor masses allowing these cells to be less resistant to the high concentration of drug.[18]
The major disadvantage of conventional TACE procedures is the rapid washout of the chemotherapeutic out of the tumor into the systemic circulation. On the other hand, LC Bead chemoembolization has 2 major advantages over conventional TACE. First, the drug is continuously released over a 10-12 days window providing a higher overall intratumoral drug dose over a longer time.[14] Secondly, with the continuous slow release of drug, there is less systemic toxicity and therefore less post embolization syndrome.[10,19,20]
Many times peripheral located liver masses that appear successfully embolized can return with increasing size and persistent tumor enhancement on follow-up imaging. Repeat angiograms can demonstrate peripheral tumor vascular recruitment from extrahepatic collateral suppliers prohibiting effective control of the tumor. These angiographic findings were more commonly seen in advanced stages of metastatic liver disease. Those collateral arterial feeders should be separately embolized at that time. Fortunately, a complete vascular assessment during the initial selective angiography eliminated the need for repeat studies attempting to search for new collaterals each time.
There was no intraprocedural discomfort described by the patients during the doxorubicin eluted bead embolization. However, we found 10/12 (83%) of the irinotecan patients described immediate right upper quadrant pain during intraprocedureal bead delivery. This phenomenon was rapid in onset, resolved quickly and did not recur following the procedure. This clinical response does not occur with doxorubicin eluted bead placement. If needed, patients were given intravenous analgesia intra-procedurally but no premedication protocol was developed during this study. It may relate to the faster elution of the irinotecan (approximately 4 days) from the beads as compared to doxorubicin.[21] Also, the amount of liver parenchyma being treated during the embolization frequently is more extensive due to the nature of colonic metastasis.
The study investigators routinely embolized the gastroduodenal artery (GDA) to prevent the embolics from refluxing into the arterial pathways leading into the duodenum and pancreas. In this study, there was no non target duodenal, gallbladder or pancreatic embolization complications. At this institution, GDA embolization is performed in every case because of that small chance of complications related to embolization of non target vascularity. We understand that gastroduodenal artery embolization is not the standard of practice in many centers despite the use of microcatheters for delivery of the embolic material. However, we believe that preserving the gallbladder, duodenum and pancreas from preventable non target embolization is crucial. GDA embolization is a quick and technically easy procedure to perform prior to LC Bead chemoembolization not adding much procedure time to the case.
The study is a retrospective investigation of this institution’s LC Bead chemoembolization practice and there are several study limitations. First, doxorubicin was the only chemotherapeutic agent used on HCC. However, the use of irinotecan for colon metastasis was chosen based on the oncologic data at that time. The literature described irinotecan as very effective to colon metastasis when given intravenously. Therefore, the investigators used this drug initially on all colorectal metastasis patients. Unfortunately, during the early part of the study, this drug was found to be ineffective on the first 12 colon metastasis patients with a poor response given intra-arterially. From that point on, doxorubicin was used exclusively during the remaining part of the study. The reason to switch from irinotecan to doxorubicin was based solely on its poor response in the first 12 patients. Once switched, there were statistically improved results using doxorubicin compared to irinotecan on colon metastasis patients. Secondly, the authors used FDG PET-CT for their follow up imaging. MRI with dedicated liver agents have become readily available and considered sufficiently sensitive for routine use for detection of HCC which may not have been identified on follow-up FDG PET-CT. Lastly, this study consisted of a very small sample size at a single institution and may not be reflective of a larger population. However, these results were compelling and suggest the need for additional systematic or randomized studies that compare these different treatment options.
This retrospective study evaluated and compared how HCC and colorectal metastasis responded to doxorubicin and irinotecan. It demonstrated that doxorubicin eluted bead embolization resulted in longer patient survival as compared to conventional therapies previously reported in the literature.[6-9] Although the patient sample size was small, 81.8% of the HCC patients and 77% of the colon metastasis patients had either complete or partial response.
Patients receiving irinotecan had a statistically significant poor response as compared to doxorubicin. Furthermore, those patients with colorectal metastases who did not respond to irinotecan initially could be candidates for repeat embolization with doxorubicin and could hopefully improve following repeat treatment outside of this study. Overall, HCC and colon metastasis patients demonstrated the effectiveness of DEB with 91% of the HCC patients alive 24 months after treatment. Prospective randomized trials would be helpful for further evaluation in a large subset of patients. Endovascular specialists should be aware of the benefits LC Bead embolization can bring to the oncology community as malignant neoplasms of the liver continue to increase in the future.
Authors’ contributions
Guarantor of integrity of the entire study, study concepts and design, literature research, clinical studies, and manuscript preparation: G.S. Stambo
Statistical analysis: D. Cragun
Experimental studies/data analysis, and manuscript editing: G.S. Stambo, D. Cragun
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
Each patient was informed of the study and gave their consent.
Ethics approval
This was a retrospective study and did not require Institutional Review Board approval.
REFERENCES
1. Serrablo A, Tejedor L, Ramia JM. Liver Metastases - Surgical Treatment. In: Reeves H, editor. Liver Tumors - Epidemiology, Diagnosis, Prevention and Treatment. Rijeka, Croatia: InTech; 2013.
2. Sasson AR, Sigurdson ER. Surgical treatment of liver metastases. Semin Ocol 2002;29:107-18.
3. Singletary SE, Walsh G, Vauthey JN, Curley S, Sawaya R, Weber KL, Meric F, Hortobágyi GN. A role for curative surgery in the treatment of selected patients with metastatic breast cancer. Oncologist 2003;8:241-51.
4. Brown DB, Geschwind JFH, Soulen MC, Millward SF, Sacks D. Society of Interventional Radiology position statement on chemoembolization of hepatic malignancies. J Vasc Interv Radiol 2006;17:217-23.
5. Lencioni R, Crocetti L, De Simone P, Filipponi F. Loco-regional interventional treatment of hepatocellular carcinoma: techniques, outcomes, and future prospects. Transpl Int 2010;23:698-703.
6. Stambo GW, Guiney MJ. Hepatic angiosarcoma presenting as an acute intraabdominal hemorrhage treated with transarterial chemoembolization. Sarcoma 2007;2007:90169.
7. Aliberti C, Benea G, Tilli M, Fiorentini G. Chemomembolization (TACE) of unresectable intrahepatic cholangiocarcinoma with slow-release doxorubicin-eluting beads: preliminary results. Cardiovasc Intervent Radiol 2008;31:883-8.
8. Steward MJ, Warbey VS, Malhotra A, Caplin ME, Buscombe JR, Yu D. Neuroendocrine tumors: role of interventional radiology in therapy. Radiographics 2008;28:1131-45.
9. Varela M, Real MI, Burrel M, Forner A, Sala M, Brunet M, Ayuso C, Castells L, Monta-á X, Llovet JM, Bruix J. Chemoembolization of hepatocellular carcinoma with drug eluting beads: efficacy and doxorubicin pharmacokinetics. J Hepatol 2007;46:474-81.
10. Liapi E, Lee KH, Georgiades CC, Hong K, Geschwind JF. Drug-eluting particles for interventional radiology. Tech Vasc Interv Radiol 2007;10:261-9.
11. Aliberti C, Tilli M, Benea G, Fiorentini G. Trans-arterial chemoembolization (TACE) of liver metastases from colorectal cancer using irinotecan-eluting beads: preliminary results. Anticancer Res 2006;26:3793-5.
12. Jakub W, Philippe LP. Using epirubicin-loaded DC Beads® for superselective embolisation of liver tumours - initial experiences. Eur Oncol 2008;4:72-6.
13. Malagari K, Chatzimichael K, Alexopoulou E, Kelekis A, Hall B, Dourakis S, Delis S, Gouliamos A, Kelekis D. Transarterial chemoembolization of unresectable hepatocellular carcinoma with drug eluting beads: results of an open-label study of 62 patients. Cardiovasc Intervent Radiol 2008;31:269-80.
14. Lewis AL, Gonzalez MV, Lloyd AW, Hall B, Tang Y, Willis SL, Leppard SW, Wolfenden LC, Palmer RR, Stratford PW. DC bead: in vitro characterization of a drug-delivery device for transarterial chemoembolization. J Vasc Interv Radiol 2006;17:335-42.
15. Aliberti C, Tilli M, Benea G, Fiorentini G. Trans-arterial chemoembolizaiton (TACE) of liver metastases from colorectal cancer using irinotecan-eluting beads: preliminary results. Anticancer Res 2006;26:3793-5.
16. Taylor RR, Tang Y, Gonzalez MV, Stratford PW, Lewis AL. Irinotecan drug eluting beads for use in chemoembolization: in vitro and in vivo evaluation of drug release properties. Eur J Pharm Sci 2007;30:7-14.
17. Fiorentini G, Aliberti C, Turrisi G, Del Conte A, Rossi S, Benea G, Giovanis P. Intraarterial hepatic chemoembolization of liver metastasis from colorectal cancer adopting irinotecan-eluting beads: results of a phase II clinical study. In Vivo 2007;21:1085-91.
18. Lencioni R, Crocetti L, Petruzzi P, Vignali C, Bozzi E, Della Pina C, Bargellini I, Cioni D, Oliveri F, De Simone P, Bartolozzi C, Brunetto M, Filipponi F. Doxorubicin-eluting bead-enhanced radiofrequency ablation of hepatocellular carcinoma: a pilot clinical study. J Hepatol 2008;49:217-22.
19. Spada F, Fazio N, Bonomo G, Monfardini L, Vigna PD, Radice D, Boselli S, Orsi F. Hepatic intra-arterial chemotherapy in patients with advanced primary liver tumours. Ecancermedicalscience 2012;6:280.
20. Lammer J, Malagari K, Vogl T, Pilleul F, Denys A, Watkinson A, Pitton M, Sergent G, Pfammatter T, Terraz S, Benhamou Y, Avajon Y, Gruenberger T, Pomoni M, Langenberger H, Schuchmann M, Dumortier J, Mueller C, Chevallier P, Lencioni R; PRECISION V Investigators. Prospective randomized study of doxorubicin-eluting-bead embolization in the treatment of hepatocellular carcinoma: results of the PRECISION V study. Cardiovasc Intervent Radiol 2010;33:41-52.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Stambo GW, Cragan D. Response rates of hepatocellular carcinoma and hepatic colorectal cancer metastases to drug eluting bead regional liver therapy. Hepatoma Res 2017;3:141-8. http://dx.doi.org/10.20517/2394-5079.2017.12
AMA Style
Stambo GW, Cragan D. Response rates of hepatocellular carcinoma and hepatic colorectal cancer metastases to drug eluting bead regional liver therapy. Hepatoma Research. 2017; 3: 141-8. http://dx.doi.org/10.20517/2394-5079.2017.12
Chicago/Turabian Style
Stambo, Glenn W., Deborah Cragan. 2017. "Response rates of hepatocellular carcinoma and hepatic colorectal cancer metastases to drug eluting bead regional liver therapy" Hepatoma Research. 3: 141-8. http://dx.doi.org/10.20517/2394-5079.2017.12
ACS Style
Stambo, GW.; Cragan D. Response rates of hepatocellular carcinoma and hepatic colorectal cancer metastases to drug eluting bead regional liver therapy. Hepatoma. Res. 2017, 3, 141-8. http://dx.doi.org/10.20517/2394-5079.2017.12
About This Article
Copyright
Data & Comments
Data

 Cite This Article 2 clicks
Cite This Article 2 clicks










Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.