Protective effects of Amaranthus hybridus against aflatoxin B1 and fumonisin B1-induced genotoxicity in H4IIE-luc cells
Abstract
Aim: Protective effects of aqueous extract of Amaranthus hybridus against aflatoxin B1 (AFB1) and/or fumonisin B1 (FB1) on the H4IIE-luc cell line were determined by use of the methyl thiazol tetrazolium viability assay and disruption of DNA integrity.
Methods: H4IIE-luc cells were incubated with different concentrations of AFB1 and/or FB1 for 24 and 48 h with or without aqueous extract of A. hybridus.
Results: AFB1 decreased viability of cells after 24 and 48 h of exposure. EC50 values for AFB1 were 10.5 and 1.8 µmol/L for the two periods respectively. When the 48 h exposure to mycotoxin repeated with a pre-treatment of 20 and 40 µg/mL extract of A. hybridus, the EC50 changed to 3.88 and 7.67 µmol/L, respectively. H4IIE-luc cells exposed to FB1 for 24 h responded more than those incubated for 48 h. Cells treated with a combination of AFB1 and FB1 were less viable with a significant decrease at the greater concentration. The mixture of AFB1 and FB1 resulted in a significant threat to H4IIE-luc as indicated by the absence or appearance of new bands in random amplified polymorphic DNA analysis, which demonstrated damage to DNA. The protective effects were probably due to greater content of total phenolics, carotenoids, β-carotene, folic-, linolenic-, linoleic and palmitic acids, as well as calcium, magnesium, iron, zinc, and selenium observed in the extract.
Conclusion: Exposure to 40 μg/ml of extract of A. hybridus protected cells from damage to DNA by stabilizing DNA.
Keywords
Introduction
Mycotoxins are secondary metabolites of fungi which are associated with certain disorders in animals and humans. Contamination of grains by mycotoxins is a worldwide problem affecting staple crops such as corn (maize) and small grains (such as wheat), as well as tree nuts, peanuts, sorghum, and others.[1] Some mycotoxins are now linked with the incidence of certain types of cancer and it is this aspect that has evoked global concern over feed and food safety.[2] Aflatoxins (AFs), ochratoxins, trichothecenes, zearelenone, fumonisins (FBs), tremorgenic toxins, and ergot alkaloids are the mycotoxins of greatest agro-economic importance[3] and are known to be hepatotoxic, genotoxic, immunosuppressive, nephrotoxic, teratogenic, and carcinogenic.[4] The Food and Agricultural Organization of the United Nations has estimated that up to 25% of the world’s food crops are significantly contaminated with mycotoxins.[5] However, in Africa, the presence of mycotoxins in food is often overlooked due to the population’s ignorance, lack of regulatory mechanisms, poor facilities for storing large volumes of food products, and the introduction of contaminated commodities into the human food chain during chronic food shortage caused by droughts, wars, and political and economic instability.[6]
At present, the interactions between AFB1 and FB1 with regard to their toxic and carcinogenic properties were discussed in several reports. A synergistic effect between exposure to mycotoxins and some important diseases in Africa, such as malaria, kwashiorkor, liver cancer and human immunodeficiency virus (HIV)/acquired immune deficiency syndrome has been suggested.[7] Concerns about mycotoxins has increased during the last few decades because of their implications to human and animal health and productivity, as well as the economics of their management, and how they influence international trade.[8] This has led to the development of maximum tolerated limits for mycotoxins in various countries. The European Union has legislated maximum permitted levels of 2 ng/g dry mass for AFB1 and 4 ng/g for total AFs (B1, B2, G1, and G2) in various products.[9] Considering the extremely potent carcinogenicity of aflatoxins, most developed nations regulate limits of AFs as small as reasonably achievable. Several studies have shown that AFB1 and FB1 are cytotoxic, inhibiting the viability of different cellular models, mostly liver and kidney cells.[10-16]
The Amaranthus plant has been used extensively in rural South Africa as a traditional food and is commonly known as morogo. Amaranthus species are good sources of β-carotene, polyphenols, vitamin C, calcium and iron.[17,18] Moreover, a joint publication of the United Nations Development Program and the Food and Agriculture Organization, expressed the view that wild-growing food plants are an affordable and practical source of nutrition to improve the nutritional status of rural HIV-affected households.[19] The present study was carried out to assess whether the aqueous extracts of A. hybridus can protect rat hepatoma cells against FB1 and AFB1 induced cytotoxicity and disruption of DNA integrity.
Methods
Chemicals
FB1 and AFB1 (98% purity) and other standards were purchased from Sigma Chemicals Co (St. Louis, MO, USA). The DNA extraction kit (DNeasy blood and tissue kit) was obtained from Qiagen (Hilden, Germany). The one hundred base pair DNA ladder, polymerase chain reaction (PCR) master mix and DNase/RNase free water were obtained from Fermentas Inc. (Glen Burnie, Maryland, USA). Supertherm Taq polymerase was purchased from JM Holding (UK). Forty primers were obtained from Operon Technologies (Alameda, CA, USA). All solvents were of analytical grade and were purchased from Burdick and Jackson (Muskegon, MI, USA).
Plant materials
Stems and leaves of A. hybridus were collected from a residential garden in the city of Potchefstroom, North-West Province, South Africa. The plant material was freeze-dried and pulverized and 1 g dry mass of the lyophilized plant powder was infused with 10 mL water for 24 h at room temperature. After centrifugation, the supernatant was freeze-dried and stored at 4 °C until used.
Determination of chemical composition of the extract
Extraction of total phenolic contents in the plant was carried out in triplicate, according to the modified method of van der Walt et al.[18] and Kähkönen et al.[20] Total phenolic content was expressed as mean ± standard deviation (SD) and gallic acid equivalents was expressed in mg/100 g dm.
Total carotenoid content for the plant was extracted and analysed in triplicate as described by Edwards et al.[21] and modified by van der Walt et al.[18] Total carotenoid content is presented as mean ± SD in mg/100 g dm.
Quantification of beta-carotene
Beta-carotene was extracted according to the modified method described by Lakshiminarayana et al.[22] and was quantified by high pressure liquid chromatography coupled to a photodiode array detector capable of ultraviolet-visible absorption spectrum according to the methods of de Ancos et al.[23] The content was expressed as mean ± SD in mg/100 g dm.
Folic acid
Folic acid was quantified at the South African Bureau of Standards (Pretoria, South Africa). A standard method for the microbiological assay of folic acid in foods and pharmaceutical products was followed according to Barton-Wright[24] and AOAC.[25]
Quantification of fatty acids
Fatty acids were identified and quantified by use of gas chromatography coupled with mass spectrometry system with split-less injection. An Agilent 6890 gas chromatograph ported to a 5973 mass selective detector (CA, USA) was used according to the method described by van der Walt et al.[17]
Mineral and trace element analysis
Minerals and trace elements were quantified by use of an Agilent 7,500 inductively coupled argon plasma mass spectrometer as described by van der Walt et al.[18] Three separate samples were analysed and values were reported as the mean ± SD in mg/100 g dm.
Cytotoxicity measurements
The mammalian model was rat hepatoma cells (H4IIE-luc) that had been stably transfected with a firefly luciferase reporter gene under control of the dioxin response element and thus the aryl hydrocarbon receptor mechanism.[26] These cells had originally been developed as a reporter gene assay to detect and semi-quantify the levels of certain groups of persistent organic pollutants.[26] Since these cells are essentially still mammalian cells, they were useful to assess whether extracts of selected A. hybridus can be protective against AFB1 and FB1 or their mixture.
Cells were seeded with a density of 1.0 × 104 cells/mL media in the inner 60 wells of a 96-well microplate. Growth medium was Dulbecco’s Modified Eagle’s Medium (Sigma, D2902) supplemented with 0.044 mol/L NaHCO3 and 10% foetal bovine serum (Gibco). The volume in each well was 250 µL. The outer cells received 250 µL Dulbecco’s phosphate buffered saline (PBS) to create a homogenous microclimate across all wells containing cells and incubation conditions were 37 °C in a humidified 5% CO2: air mixture. The plates were seeded and after an initial 24 h incubation medium was replaced with medium containing varying concentrations of AFB1 (50, 25, 2.5, 0.25, 0.025 µmol/L ) and FB1 (200, 100, 10, 1, 0.1 µmol/L) dissolved in methanol. A combination of the already mentioned concentrations of AFB1 and FB1 were also tested: 50 µmol/L AFB1 plus 200 µmol/L FB1; 25 µmol/L AFB1 plus 100 µmol/L FB1 and so on. Two exposure periods 24 h and 48 h were investigated. In order to evaluate the protective effect of extracts of A. hybridus, this experiment was repeated with the following adjustments: after the initial 24 h incubation period the media was replaced with media containing either 20 or 40 µg/mL A. hybridus and incubated for another 24 h which was followed by the mycotoxin exposure routine, but for only the 48 h period. The controls included (1) 11 wells with cells and nutrient medium only for the duration of the entire experiment (when media was replaced, their media was replaced with fresh nutrient medium) and (2) 6 wells with cells and plant extract containing media only. The mycotoxin exposures were dosed in triplicate.
The viability of H4IIE-luc cells was determined using the methyl thiazol tetrazolium (MTT) salt assay in which the mitochondria of live cells metabolise the yellow MTT into blue formazan.[27] A final concentration of 500 µg/mL MTT was incubated for 30 min and blue formazan crystals dissolved with dimethyl sulfoxide. The absorbance was measured spectrophotometrically at 560 nm. The amount of formazan gives an estimation of the proportion of viable cells. The percentage of viable to dead cells was calculated by comparison with a control (untreated and solvent control). The MTT assay assessed the viability of H4IIE-luc cells that were subjected to the two A. hybridus extract concentration treatments compared to viability of cells that were not treated with A. hybridus extracts prior to mycotoxin exposure.
DNA extraction
Cells were harvested by first washing away non-adherent dead cells with PBS before trypsinising (0.25% trypsin, 0.1% versene ethylenediaminetetraacetic acid) adherent cells. Enzyme activity was stopped by the addition of media. The cell suspension was centrifuged for 5 min (300 g) at room temperature. The genomic DNA was extracted from the cells according to the Qiagen instruction manual. The concentration of DNA was determined by photometry (NanoDrop ND-1000 Spectrophotometer) and the purity of the DNA was judged by examining the ratio of absorbency at 260/280 nm.[28]
Random amplified polymorphic DNA-polymerase chain reaction analysis
Amplification of DNA fragments was carried out on an ICycler (Bio-Rad, UK) thermal cycler using 20 primers purchased from the Operon Biotechnologies (BioCampus Colonge Nattermannalle, Germany). PCR amplification was conducted in a 25 µL reaction volume containing 10 ng genomic DNA, 12.5 pmol Master mix (2X) (Fermentas life science, USA), 1.0 units of Supertherm Taq polymerase and 50 pmol primer. PCR reactions were carried out in a thermocycler (BIO RAD C1000) programmed with initial denaturation period for 5 min at 95 °C, followed by 40 cycles denaturation (95 °C for 30 s), primary annealing at 37 °C for 1 min and extension at 72 °C. Amplification was terminated by a final extension period of 72 °C for 5 min. Reaction products were stored at -80 °C prior to electrophoresis.
Gel electrophoresis
Amplified products together with marker (100 bp DNA) were resolved by gel electrophoresis 60 V/cm for 135 min on 2% agarose gel in TAE buffer containing 0.001 mg/mL ethidium bromide. Gels were photographed by a Gel Documentation system (Gensnap) equipped with its software (Synegen, UK).
Band analysis
Gels of control and exposed DNA samples were run for each of the 20 primers [Table 1]. A DNA ladder of 100 bps was also run in each gel. Bands in PCR products were analysed by TotalLab Quant (V11.5:TL100-LX59-7YF4-EX). The fluorimetric profiles of each amplification reaction were studied both qualitatively and quantitatively by comparing profiles from the control and DNA exposed to the extracts. Each change observed in random amplified polymorphic DNA (RAPD) profiles of the treated groups (disappearances and appearance of bands in comparison to the control RAPD profiles) was given the arbitrary score of +1. The average was then calculated for each experimental group exposed to the mycotoxins for varying time periods. Genomic stability (%) was calculated as “100 - (100 a/n)” where “a” is the average number of changes in DNA profiles and “n” is the number of bands selected in control DNA profiles.[29]
Sequences of the primers used to amplify cell line of hepatoma (H4IIE-luc) cells
| Primer | Sequence 5’-3’ | Primer | Sequence 5’-3’ |
|---|---|---|---|
| D01 | ACCGCGAAGG | D11 | AGCGCCATTG |
| D02 | GGACCCAACC | D12 | CACCGTATCC |
| D03 | GTCGCCGTCA | D13 | GGGGTGACGA |
| D04 | TCTGGTGAGG | D14 | CTTCCCCAAG |
| D05 | TGAGCGGACA | D15 | CATCCGTGCT |
| D06 | ACCTGAACGG | D16 | AGGGCGTAAG |
| D07 | TTGGCACGGG | D17 | TTTCCCACGG |
| D08 | GTGTGCCCCA | D18 | GAGAGCCAAC |
| D09 | CTCTGGAGAC | D19 | CTGGGGACTT |
| D10 | GGTCTACACC | D20 | ACCCGGTCAC |
Statistical analysis
Values for EC50 and cell viability were statistically analyzed with the Graphpad Prism 4.02 Inc., (La Jolla, CA, USA). Significance of differences among treatment groups was determined with the Waller-Duncan k-ratio.[30] All statements of significance were based on a probability of P < 0.05.
Results
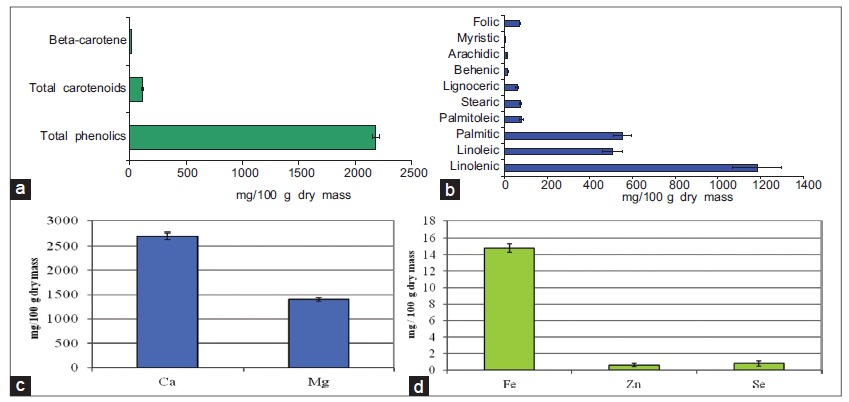
The extract was rich in polyphenols (total phenolic contents: 2181.2 mg/100 g dm, total carotenoids (113.6 mg/100 g dm) and β-carotene (18.4 g/100 g dm) [Figure 1a]. The results of the lipid profile showed significant amounts of the fatty acids, linolenic, linoleic and palmitic [Figure 1b]. The extract had moderate concentrations of palmitoleic, stearic, and lignoceric acid whereas; behenic, arachidic, and myristic acid were found in low concentrations. The extract was rich in folic acid (72 mg/100 g dm), calcium, magnesium [Figure 1c], iron, zinc and selenium [Figure 1d].
Figure 1. (a) Total phenolics, total carotenoids, and β-carotene content; (b) fatty acid profiles and folic acid content; (c) calcium and magnesium concentration; and (d) trace elements (iron, zinc, and selenium) concentration of Amaranthus hybridus extract
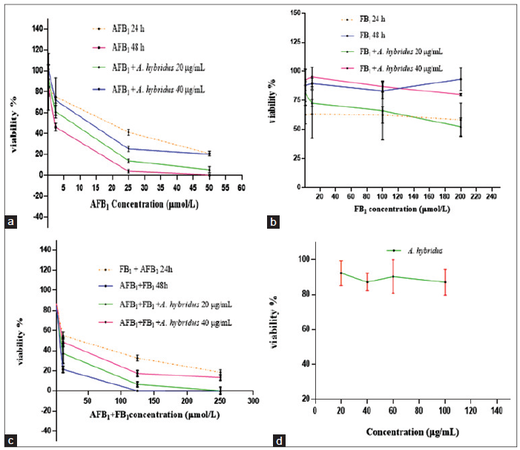
Cytotoxicity of AFB1, FB1 and mixture with or without the extract of A. hybridus on H4IIE-luc cell line as measured by the tetrazolium dye-based MTT assay are shown in Figure 2a-d. There was a significant difference in viability between cells treated with 20 µg/mL A. hybridus and those not treated before exposure to AFB1 for 48 h [Table 2]. Other combinations did not have any statistically significant difference. Percentage inhibition of the cells incubated for 24 h with FB1 showed more cytotoxicity than those incubated for 48 h. On the other hand, FB1 at the concentration of 200 µmol/L decreased cell viability to 41.6% [Figure 2b]. The protective effect of 20 μg/mL A. hybridus extract was decreased by increasing FB1 dose to 100 µmol/L [Figure 2b]. A. hybridus extract at 40 μg/mL was more efficient at protection against all concentrations of FB1.
Summary of the P values of the Wilcoxon matched pair tests to compare viability of cells exposed to FB1, AFB1, and their mixture and those treated with A. hybridus extracts prior to 48 h mycotoxin exposure
| Mycotoxins | A. hybridus extract | |
|---|---|---|
| 20 µg/mL | 40 µg/mL | |
| FB1 | 0.04* | 0.69 |
| AFB1 | 0.5 | 0.08 |
| FB1 + AFB1 | 0.69 | 0.2 |
Figure 2. The cytotoxic effects of AFB1 at different concentrations (μmol/L) without/with Amaranthus hybridus extract: (a) FB1 at different concentrations (μmol/L) without/with Amaranthus hybridus; (b) AFB1 plus with FB1 without/with Amaranthus hybridus; (c) Amaranthus hybridus extract only; and (d) on proliferation of H4IIE-luc cell line determined by MTT bioassay. FB1: fumonisin B1; AFB1: aflatoxin B1; MTT: methylthiazol tetrazolium
Overall, AFB1 was more cytotoxic than FB1 for both exposure periods [Figure 2a and b]. Exposure of H4IIE-luc cells to AFB1 led to a dose-and time-dependent decrease in cell viability. At 25 µmol/L AFB1 viability was inhibited to 58.7% and 96.1% for the 24 and 48 h exposure periods respectively. Pre-treating the cells to 40 µg/mL A. hybridus had a more protective effect than pre-treatment of 20 µg/mL [Figure 2a].
The combination of AFB1 and FB1 was more cytotoxic than AFB1 alone which indicated that FB1 increased the cytotoxicity. This was true for both exposure periods [Figure 2a-c]. However, this general trend was not corroborated by the EC50 values (EC50 = concentration by which viability was declined to 50%) [Table 3]. They were in fact slightly greater for the combined mycotoxins than exposure to AFB1 alone, meaning that 50% effect was reached at a greater mycotoxin concentration. Extract of A. hybridus alone (20-100 µg/mL) had no significant influence on viability of cells [Figure 2d].
The EC50-values of AFB1 and/or FB1 alone or in combination with A. hybridus at two exposure periods using the H4IIE-luc cell line
| Mycotoxin/plant extract | Pre-treatment concentration of A. hybridus (μg/mL) | Mycotoxin exposure time (h) | EC50 (μmol/L) |
|---|---|---|---|
| FB1 | - | 24 | ND |
| - | 48 | ND | |
| 20 | 48 | ND | |
| 40 | 48 | ND | |
| AFB1 | - | 24 | 10.55 |
| - | 48 | 1.84 | |
| 20 | 48 | 3.88 | |
| 40 | 48 | 7.67 | |
| FB1 + AFB1 | - | 24 | 24.02 |
| - | 48 | 5.86 | |
| 20 | 48 | 7.30 | |
| 40 | 48 | 14.00 | |
| A. hybridus | 5592 μg/mL |
EC50 values for AFB1 were 10.5 and 1.8 µmol/L after 24 and 48 h of exposure respectively. When the plant extract was added at 20 and 40 µg/mL, the EC50 values were 3.88 and 7.67 µmol/L after 48 h of exposure respectively. On the other hand, the EC50 for the combined mycotoxins (AFB1 + FB1) was 24.02 and 5.86 µmol/L after 24 and 48 h respectively. While the 50% inhibition of proliferation for AFB1 plus FB1 with plant extract were 7.30 and 14.0 µmol/L after the addition of 20 or 40 µg/mL A. hybridus extract, respectively. No discernible cytotoxicity was observed in cells, based on the MTT assay, when cells were exposed to FB1 at lesser concentrations. Whereas, at greater concentrations sufficient cell mortality was exhibited in the MTT assay that indicate cytotoxicity at dosages of 200 µmol/L but the quantities tested were still substantially less than those required to obtain EC50.
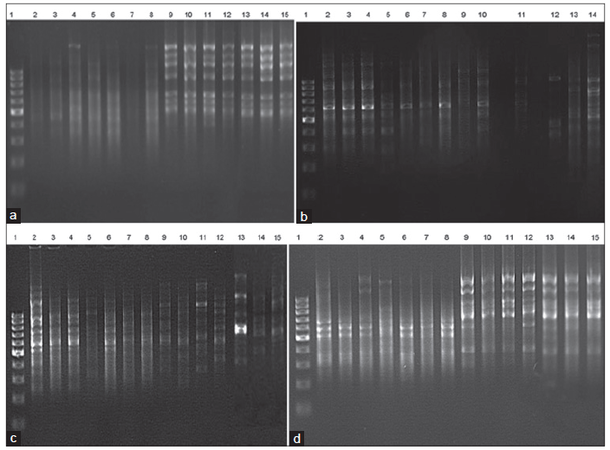
Genetic variability among treated cells was evaluated using ten oligonucleotide primers. Only five primers, D07, D09, D13, D15, and D16, gave positive and detectable bands [Figure 3] since they amplified a total of 69 different bands ranging from 144 to 2000 bp. All 69 bands were “polymorphic” given 100% polymorphism for control cells, FB1, AFB1, AFB1 plus FB1, plant extract at 40 μg/mL, 40 μg/mL plant extract plus FB1, 40 μg/mL extract plus AFB1 and 40 μg/mL extract plus 0.025 and 50 μmol/L AFB1, respectively, for all primers used. Of the 69 scorable bands, 18 (26%) were similar “monomorphic” to the control and the 40 μg/mL A. hybridus treatment (D09-700, D09-525, D09-363, D13-363, D15-1080, D15-869, D15-646, D15-547, D15-447, D15-325, D15-229, D15-176, D16-183, D16-1267, D16-813, D16-679, D16-536 and D16-417; 1 band (1.4%) was similar for control and A. hybridus extract at 40 μg/mL in all treatments after the addition of the extract at 40 μg/mL for all treatments (D16-646).
Figure 3. (a-d) RAPD profi les of genomic DNA from cell line of hepatoma (H4IIE-luc) of rats following exposure to FB1 and/or AFB1 for various time periods. Four pictures represents PCR products with primer OPD07 (a), OPD09 (b), OPD15 (c), and OPD16 (d), respectively, at less and greater concentration. The DNA marker (100 bp) in lanes 1 and 2 represents cells only; lane 3 represents cells plus FB1 (0.1 μmol/L); lane 4 represents cells plus FB1 (200 μmol/L); lane 5 represents cells plus AFB1 (0.025 μmol/L); lane 6 represents cells plus AFB1 (50 μmol/L); lane 7 represents cells plus the mixture of FB1 and AFB1 (0.1 + 0.025 μmol/L); lane 8 represents cells plus the mixture of FB1 and AFB1 (200 + 50 μmol/L); lane 9 represents cells plus Amaranthus hybridus extract (40 μg/mL); lane 10 represents A. hybridus extract(40 μg/mL) plus FB1 (0.1 μmol/L); lane 11 represents Amaranthus hybridus extract (40 μg/mL) plus FB1 (200 μmol/L); lane 12 represents Amaranthus hybridus extract (40 μg/mL) plus AFB1 (0.025 μmol/L); lane 13 represents Amaranthus hybridus extract (40 μg/mL) plus AFB1 (50 μmol/L); lane 14 represents Amaranthus hybridus extract (40 μg/mL) plus the mixture of FB1 and AFB1 (0.1 + 0.025 μmol/L), and lane 15 represents Amaranthus hybridus extract (40 μg/mL) plus the mixture of FB1 and AFB1 (200 + 50 μmol/L). FB1: fumonisin B1; AFB1: aflatoxin B1; RAPD: random amplification of polymorphic DNA; PCR: polymerase chain reaction
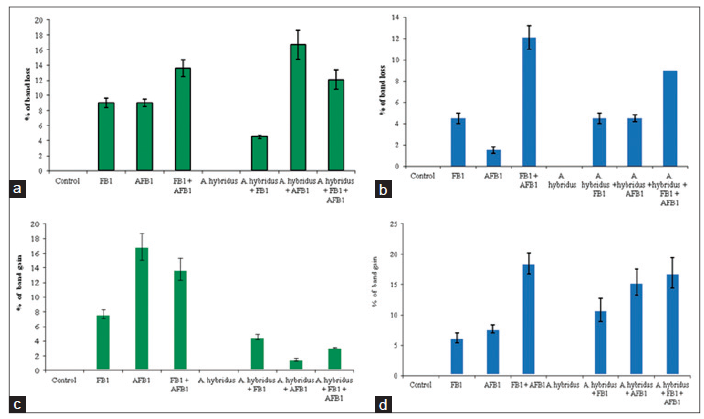
Quantitative analysis of these bands, expressed as a percentage of band loss, shows a time-dependant relationship. The increase in band loss is related to the increase in time period [Table 3; Figure 4]. Similarly, in case of band loss at the short exposure period (24 h), 26 out of 66 bands (39.4%) disappeared [Figure 4a]. At the 48 h exposure period, 44 out of 65 bands vanished which representing 75.4% [Figure 4b]. A protective effect of the extract at 40 μg/mL was observed for the short exposure period and 14 out of 69 bands (20.29%) disappeared, compared to the 28 out of 69 bands (40.58%) for the longer period which disappeared.
Figure 4. Genomic damage. The percentage of altered bands in each treatment of low and high concentration of FB1 and AFB1 detected by RAPD-PCR. (a) Average band loss after 24 h; (b) average band loss after 48 h; (c) average band gains after 24 h; and (d) average band gain after 48 h. FB1: fumonisin B1; AFB1: aflatoxin B1; RAPD: random amplification of polymorphic DNA; PCR: polymerase chain reaction
Meanwhile, bands also appeared at the short exposure period, 43 new bands out of 66 bands were amplified which represents 65.2%. In the same trend, at the longer exposure period, 31 out of 65 appeared which represents 47.7% [Figure 4c]. The protective effect of the 40 μg/mL plant extract was observed as the production of new bands appeared at the short exposure period (24 h) since 22 out of 66 bands emerged which represents 33.3% while; at the long exposure period, 6 out of 66 bands occurred and represent 9.09 % [Figure 4d].
Profiles of RAPD-PCR and the number of bands that appeared or disappeared in the DNA of hepatoma H4IIE-luc at various exposure periods are shown in Table 4. A maximum of 9 bands vanished in the mixture of FB1 at 200 μmol/L and AFB1 at 50 μmol/L plus the plant extract-treated cells for 48 h with OPD 15 primer. Whereas, the maximum appearance of 7 new bands were observed in the AFB1 at 50 μmol/L plus plant extract-exposed cells at 48 h with OPD 15 too.
Frequency of appearance and disappearance of bands in the RAPD profi les of genomic DNA from cell line of hepatoma (H4IIE-luc) of rats following exposure to FB1 and/or AFB1 for various time periods
| Name of primer | Change in RAPD profi le | Control | FB1 | FB1 | AFB1 | AFB1 | AFB1 + FB1 | AFB1 + FB1 | AE | AE + FB1 | AE + FB1 | AE + AFB1 | AE + AFB1 | AE + AFB1 + FB1 | AE + AFB1 + FB1 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (LC) | (HC) | (LC) | (HC) | (LC) | (HC) | (LC) | (HC) | (LC) | (HC) | (LC) | (HC) | ||||
| D7 | A | 0 | 3 | 5 | 4 | 4 | 0 | 6 | 0 | 0 | 2 | 1 | 0 | 1 | 0 |
| D | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | |
| D9 | A | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 5 | 0 |
| D | 0 | 0 | 0 | 1 | 3 | 3 | 2 | 0 | 0 | 6 | 3 | 0 | 0 | 2 | |
| D13 | A | 0 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 1 | 0 | 3 | 0 | 2 | 1 |
| D | 0 | 0 | 3 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | |
| D15 | A | 0 | 0 | 0 | 1 | 4 | 6 | 3 | 0 | 2 | 1 | 7 | 0 | 0 | 0 |
| D | 0 | 3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 | 6 | 9 | |
| D16 | A | 0 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| D | 0 | 0 | 0 | 0 | 2 | 5 | 8 | 0 | 2 | 1 | 2 | 3 | 0 | 0 |
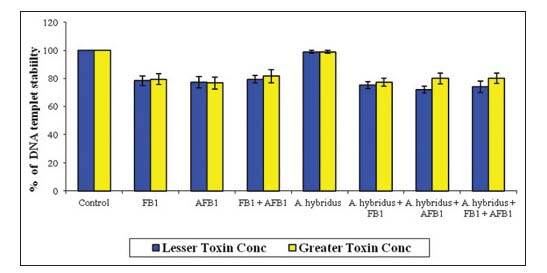
The percentage of DNA template stability in the treated cells in comparison to the controls at various concentrations is presented in Figure 5. The results showed that there was a significant difference in the DNA template stability between the control and all the treated groups, no significant difference was observed in the DNA template stability between the control and cells treated with 40 μg/mL A. hybridus extract. The protective effect of the extract at 40 μg/mL appeared as marked in the stability of DNA in all treatments.
Discussion
Polyphenols are a class of phytochemicals that contribute to total antioxidant capacity of dark green leafy vegetables.[31] They have aromatic rings, and achieve their antioxidant activities mainly through donation of hydrogens.[32] In the current study, A. hybridus extract was found to be enriched in phenolic compounds in amounts comparable to those of conventional and commercially-grown non-conventional vegetables.[17] Total phenolic concentrations reported herein were similar to those reported in commercial spinach.[33] Total phenolic concentrations in leaves of commercially-produced Ipomaea batata, which is also eaten as morogo in South Africa were similar.[34]
Carotenoids are pigment molecules responsible for the color of many fruits and vegetables, have important functions in photosynthesis and are abundant in plant leaves. Carotenoid and beta-carotene concentrations reported in the current study were comparable with that of baby spinach reported previously.[35] Bioavailability of carotenoids in dark green leafy vegetables is reduced by the leaf matrix.[36] Notwithstanding this limitation, and distinct from being Vitamin A precursors, carotenoids also exhibit considerable antioxidant capacity based on their symmetrical linear 40-carbon tetraterpene structure, which features alternating double and single carbon-carbon bonds.[23,34] Folic acid concentration in A. hybridus reported in the current study was similar to that reported previously in African vegetables.[18] In the present study, six saturated fatty acids, one monounsaturated fatty acid and two poly-saturated fatty acids were isolated. These results were in accordance with those reported by Weather[37] who suggested that dark green leafy vegetables generally contain small amounts of fat predominantly in the form of polyunsaturated fatty acids.
Calcium (Ca2+) plays a vital role in regulating cellular transmembrane trafficking of elements and molecules.[38] Dark-green leafy vegetables, therefore, are primary sources of minerals and trace elements.[39] In the present study, the extract was enriched in calcium and magnesium, and trace elements iron, zinc and selenium. Mineral and trace element content of plant leaves is a function of the environment and in leafy vegetables would be strongly influenced by the chemical composition of the soil and the climate.[40,41] The current results were similar to those reported previously,[42] that suggests that wild morogo should be considered an important source of calcium, magnesium, iron and zinc, particularly for households that are not in a position to access conventional vegetables, whether for economic or demographic reasons.
Exposure of H4IIE-luc cells to AFB1 resulted in death of cells in a concentration and time dependent manner. H4IIE-luc cells were more sensitive to AFB1, and AFB1 plus FB1 mixture compared to the control and FB1 alone. The results showed that treatment with the plant extract 24 h prior to mycotoxin exposure succeeded to blocks the AFB1 toxicity in H4IIE-luc cells line. This may be associated with the content of vitamins, antioxidants and minerals in the plant extract. This result suggested that natural vitamins, provitamins, carotenoids, chlorophyll, phenolics and synthetic compounds with antioxidant properties could potentially be effective against the toxic consequence of these mycotoxins.[43]
Toxic effects of FB1 were more pronounced after 24 h than 48 h exposure. However, the cytotoxic effect of FB1 was eliminated at lesser concentrations, suggesting the rapid metabolism of this mycotoxin.[44] These results were similar to those observed during an in vivo study that proved the elevation of sphinganine was reversible after short-term exposure.[45] Disruption of sphingolipid metabolism as a specific cytotoxic response to FB1 exposure and sphangonine reached its maximum concentration after 48 h.[46,47] Several reports indicate that FB1 inhibited cell proliferation in different cell lines H4TG, MDCK, NIH3T3, and LLC-PKI.[47,48] Among 15 mammalian cell lines, MDCK and H4TG were found to be the most sensitive to FB1 with EC50-values of 2.5 and 4 µg/mL, respectively, after 4 days exposure.[49]
AFB1 is a well-known genotoxicant able to alter the genetic constitution of an organism by inducing insults of various types. Changes in profiles were observed between control and all mycotoxin treatments. Differences in profiles of bands between the control and treated samples might be due to AFB1 and/or FB1-induced point mutations and/or base modifications elicited in the genome.[50] All primers used in this study could detect changes in all treatments which might be due to a latent phase required for the appearance of adequate number of cells with genetic damage.
Alterations observed in the present study included the absence and/or presence of bands in all treatment groups. The appearance and disappearance of bands might be associated with genetic rearrangements or clastogenic effects of the toxicant. Such alterations in the genome might subsequently interfere with binding of primers or amplification step.[51] Increases in band intensity and appearance of new PCR products has been attributed to conformational changes in DNA,[52] which might improve the access of primer(s) to the binding site(s). Furthermore, enhancement and reduction of signal intensity of an amplified DNA fragment might be related to localized over- or under-amplification of that gene locus in the genome, which could result from changes at the chromosome level.
Instability in template DNA was observed in all treatments which may be due to DNA damage. Although RAPD appears to be instrumental in observing definitive changes, it requires enough time and sufficient theoretical knowledge for initial standardization to obtain reproducible and unambiguous results. Interpretation of molecular events responsible for differences observed in the RAPD pattern is not easy, since different DNA alterations may induce similar types of changes. The RAPD is known to produce non-reproducible bands, but once established and standardized, there are certain additional benefits to using this method for early genotoxicity studies other than being fast.
Differences in sensitivity were observed, depending on the primer sequence. This observation suggests the mode of action of FB1 and/or AFB1. The five primers used showed a greater alteration after the treatment and the appearance of new bands in all the extracts-treated groups were produced from those primers. The mechanism by which these toxins affect the sequence of DNA has been extensively supported in the literature.[53] Some of the AFB1 adducts have been shown to be capable of inducing base substitution, frameshifts, insertions and deletions at specific loci of the DNA. For example, AFB1 adduct induce G > T transversion at specific loci within p53.[54,55] The resulting alterations in DNA can induce changes in the DNA sequence at specific places generating different annealing primer-template sites.[56] This is probably the reason why altered bands were always the same in most of the concentrations and the exposure periods in both the qualitative and the quantitative analysis.
Generation of new annealing primer-template sites would be in accordance with the presence of new bands in the amplification profiles. The nature of the RAPD reaction, where the final products are the result of an exponential multiplication of the most abundant and stable fragments co-amplified in the first cycles is the cause of the differences in the concordance among replicates. In other words, it is necessary that new annealing sites appear in a high proportion of the cell population to get a high reproducibility. The first new bands appeared at the high concentration of FB1 and/or AFB1 (D-71466, D-7525) suggested that the proportion of cells with a new annealing primer-template was increased at the greater concentration. The RAPD assay is able to detect mutation only if they occur in at least 2% of the DNA.[56] A concentration-dependent effect was observed when the same chemical and the same cells were used. Similar results were previously demonstrated a dose-dependent effect of genotoxic action of mycotoxin when measured by micronuclei induction.[57]
The combined use of in vitro systems and the RAPD technique permits detection of alterations in DNA caused by multiple mechanisms with a sufficient degree of sensitivity. Alterations were detected in an unspecific form by losses and/or gains of bands and variations in the amplification intensity. Nevertheless, when the objective is to establish the existence of DNA damage, that is, for hazard identification in risk assessment studies, the presence in the fingerprint of any of these abnormalities would be enough to identify a genotoxic effect. For example, the presence of one or both of the two new bands in DNA extracts of cells treated either with a chemical or with an environmental sample can be considered as a suitable genotoxicity biomarker of chronic exposure.
The protective effects of A. hybridus extract against FB1 showed that the extract was more effective at its greater dose than at the lesser dose. In addition, the ability of the extract to eliminate the cytotoxic effects induced by AFB1 appeared less effective compared that induced by FB1. The difference between AFB1 and FB1-induced cytotoxic effects may be due to the stronger oxidative stress caused by AFB1 even at a lower concentration than FB1. Several studies showed the benefits of antioxidant compounds in the diet against the toxicity of mycotoxins.[58] The inhibition of DNA and protein synthesis induced by AFB1 and FB1 were decreased by pre-treatment of the CaCo-2 and Hep G2 cell lines with the antioxidant cyaniding-3-0-β-glucopyranoside.[58] The ability of A. hybridus extract to inhibit the cytotoxic effects induced by the mixture of AFB1 and FB1 was more pronounced at the higher concentration of the extract (40 µg/mL) than the lower concentration (20 µg/mL). Moreover, this protective effect was smaller in the case of the mycotoxin mixture compared to that of FB1 only. These results were similar to those reported by Guerra et al.[59] who suggested that the inhibitory action of cyaniding-3-O-β-glucopyranoside on AFB1 and OTA-induced toxicity is likely to be attributed to its antioxidant power.
Previous studies showed that the aqueous extract of A. hybridus has a significant immune-stimulating effect and its stem extract has been credited with antimalarial activity and these effects are attributed to presence of amaranthine, isoamaranthine, hydroxycinnamates, quercetin, kaempferol glycosides, amaranthoside, amaricin and stigmasterol glycoside.[60] These authors concluded that the hepatoprotective activity of A. hybridus might be due to antioxidant defence factors and phenolics might be the main constituents responsible for activity. Isolated polyhydroxylated nerolidols which have antiradical and reducing capacities and could act as antilipoperoxidants.[61]
In conclusion, A. hybridus extract has a high content of total phenolic, total carotenoids, β-carotene, folic acid, linolenic, linoleic, palmitic, calcium, magnesium, iron, zinc and selenium. AFB1 or FB1 alone or in combination induced toxic effects on rat hepatoma cells. However, mixture of the two mycotoxins was most potent. The H4IIE-luc cells showed a weak antagonistic effect when exposed to the mixture of the two mycotixins compared to the single toxin exposures. Viability of cells was decreased by increasing concentrations of mycotoxins. Moreover, the binary mycotoxin mixture posed a significant threat to the treated hepatoma cell line as indicated by the absence/appearance of new bands beside the severe DNA damage. Combined treatment with A. hybridus extract and mycotoxins resulted in significant improvement in cell viability accompanied with a significant decrease in DNA damage and genotoxic effects. This improvement was more pronounced in the individual toxin-treated cells and in a dose dependent manner of the extract.
Financial support and sponsorship
The Morogo Research Program gratefully acknowledges the National Research Foundation of South Africa (Focus Area Grant FA2004050600064) and National Research Center, Cairo, Egypt Project #10070112 for financial support of this study. Prof. Giesy was supported by the Canada Research Chair program, a Visiting Distinguished Professorship in the Department of Biology and Chemistry and State Key Laboratory in Marine Pollution, City University of Hong Kong, the 2012 “High Level Foreign Experts” (#GDT20143200016) program, funded by the State Administration of Foreign Experts Affairs, the P.R. China to Nanjing University and the Einstein Professor Program of the Chinese Academy of Sciences.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Food and Agricultural Organization. Food Nutrition and Agriculture. Food for the Future. Rome: FAO; 1991.
2. Castegnaro M, Dcgregor D. Carcinogenic risk assessment of mycotoxins. Rev Med Vet 1998;149:671-8.
3. Richard JL. Some major mycotoxins and their mycotoxicoses - an overview. Int J Food Microbiol 2007;119:3-10.
5. World Health Organization. Basic Food Safety for Health Workers. Geneva: WHO; 1999.
6. MERCK (Medical Research Council). Aflatoxin in peanut butter. Science in Africa. Darmstadt: MERCK; 2006.
7. Wagacha JM, Muthomi JW. Mycotoxin problem in Africa: current status, implications to food safety and health and possible management strategies. Int J Food Microbiol 2008;124:1-12.
8. World Health Organization. Mycotoxins in African foods: implications to food safety and health. AFRO Food Safety Newsletter. Geneva: WHO; 2006.
9. van Egmond HP. Mycotoxins: regulations, quality assurance and reference materials. Food Addit Contam 1995;12:321-30.
10. Müller G, Burkert B, Möller U, Diller R, Rohrmann B, Rosoner H, Köhler H. Ochratoxin A and some of its derivatives modulate radical formation of porcine blood monocytes and granulocytes. Toxicology 2004;199:251-9.
11. El-Nekeety AA, El-Kholy W, Abbas NF, Ebaid A, Amra HA, Abdel-Wahhab MA. Efficacy of royal jelly against the oxidative stress of fumonisin in rats. Toxicon 2007;50:256-69.
12. Abdel-Wahhab MA, Hassan NS, El-Kady AA, Mohamed YA, El-Nekeety AA, Mohamed SR, Sharaf HA, Mannaa FA. Red ginseng protects against aflatoxin B1 and fumonisin-induced hepatic pre-cancerous lesions in rats. Food Chem Toxicol 2010;48:733-42.
13. Hassan AM, Hassan NS, Mohamed SR, El-Nekeety AA, Abdel-Wahhab MA. Aquilegia vulgaris L extract counteracts oxidative stress and cytotoxicity of fumonisin in rats. Toxicon 2010;56:8-18.
14. Hassan AM, Abdel-Aziem SH, Abdel-Wahhab MA. Modulation of DNA damage and alteration of gene expression during aflatoxicosis via dietary supplementation of Spirulina (Arthrospira) and whey protein concentrate. Ecotoxicol Environ Safety 2012;79:294-300.
15. Abdel-Aziem SH, El-Nekeety AA, Barakat IA, Mohamed IM, Abdel-Wahhab MA. Aquilegia vulgaris extract protects against the oxidative stress and the mutagenic effects of cadmium. Exp Toxicol Pathol 2011;63:337-44.
16. Abdel-Aziem SH, Hassan AM, Abdel-Wahhab MA. Dietary supplementation with whey protein and ginseng extract counteract the oxidative stress and DNA damage in rats fed aflatoxins-contaminated diet. Mutat Res 2011;723:65-71.
17. van der Walt AM, Ibrahim MI, Bezuidenhout C, Loots DT. Linolenic acid and folate in wild-growing African dark leafy vegetables (morogo). Public Health Nutr 2008;12:525-30.
18. van der Walt AM, Loots DT, Ibrahim MI, Bezuidenhout C. Minerals, trace elements and antioxidant phytochemicals in wild African dark-green leafy vegetables (morogo). South Afric J Sci 2009;105:444-8.
19. Gari JA. Plant Diversity, Sustainable Livelihoods and the HIV/AIDS Crisis. A Joint Publication of the United Nations Development Program and the Food and Agriculture Organization; 2004. Available from: http://www.hivpolicy.org/Library/HPP000946.pdf [Last accessed on 2015 Sep 16].
20. Kähkönen MP, Hopia AI, Vurela HJ, Rauha J, Pihlaja K, Kujala T, Heinonen M. Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem 1999;47:3954-62.
21. Edwards EJ, Saint RE, Cobb AH. Is there a link between greening and light enhanced glycoalkaloid accumulation in potato (Solanum tuberosum L.) tubers? J Food Sci Agric 1998;76:327-33.
22. Lakshiminarayana R, Raju R, Krishnakantha TP, Baskare V. Determination of major carotenoids in a few Indian leafy vegetables by high-performance liquid chromatography. J Agric Food Chem 2005;53:2838-42.
23. de Ancos B, Gonzalez E, Cano MP. Effect of high-pressure treatment on the carotenoid composition and radical scavenging activity of persimmon fruit purees. J Agric Food Chem 2000;48:3542-8.
24. Barton-Wright EC. Practical Methods for the Microbiological Assay of the Vitamin B-complex and Amino Acids. London: United Trade Press; 1961.
25. Association of Official Analytical Chemists. Folic acid. In: Official Methods of Analysis. 16th ed. Washington, DC: AOAC; 1999.
26. Nie M, Blankenship AL, Giesy JP. Interactions between aryl hydrocarbon receptor (AhR) and hypoxia signalling pathways. Environ Toxicol Pharmacol 2001;10:17-27.
27. Vistica DT, Skehan P, Scudiero D, Monks A, Pittman A, Boyd MR. Tetrazolium-based assays for cellular viability-a critical-examination of selected parameters affecting formazan production. Cancer Res 1991;51:2515-20.
28. Bradford MM. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248-54.
29. Atienzar FA, Jha AN. The random amplified polymorphic DNA (RAPD) assay and related techniques alied to genotoxicity and carcinogenesis studies: A critical review. Mutat Res 2006;613:76-102.
30. Waller RA, Duncan DB. A Bayes rule for the symmetric multiple comparison problems. J Am Stat Assoc 1969;64:1484-503.
31. Duthie GG, Duthie SJ, Kyle JAM. Plant polyphenols in cancer and heart disease: implications as nutritional antioxidants. Nutr Res Rev 2000;13:79-106.
32. Puoane T, Steyn K, Bradshaw D, Laubscher R, Fourie J, Lambert V, Mbananga N. Obesity in South Africa: the South African demographic and health survey. Obes Res 2002;10:1038-48.
33. Pandjaitan N, Howard LR, Morelock T, Gil MI. Antioxidant capacity and phenolic content of spinach as affected by genetics and maturation. J Agric Food Chem 2005;53:8618-23.
34. Lako J, Trenerry VC, Wahlqvist M, Wattanapenpaiboon N, Sotheeswaran S, Premier R. Phytochemical flavonols, carotenoids and the antioxidant properties of a wide selection of Fijihan fruit, vegetables and other readily available foods. Food Chem 2007;101:1727-41.
35. Bergquist S. Bioactive compounds in baby spinach (Spinacia oleracea L)- effects of pre- and postharvest factors. Ph.D. Thesis, Swedish University of Agricultural Sciences, Sweden; 2006.
36. de Pee S, Bloem MW. The bioavailability of (pro) vitamin A carotenoids and maximizing the contribution of homestead food production to combating vitamin A deficiency. Int J Vitam Nutr Res 2007;77:182-92.
37. Weathersa.co.za.Pretoria, South Africa: South African Weather Service. Available from: http://www.weathersa.co.za. [Last accessed on 2006 Sep].
38. Borek C. Cancer prevention by natural dietary antioxidants in developing countries. In: Bahorun T, Gurib-Fakim A, editors. Molecular and Therapeutic Aspects of Redox Biochemistry. London: OICA International (U.K.) Limited; 2003:259-69.
39. Van Duyn MA, Pivonka E. Overview of the health benefits of fruit and vegetable consumption for the dietetics professional: selected literature. J Am Diet Assoc 2000;100:1511-21.
40. Mnkeni AP, Masika P, Maphaha M. Nutritional quality of vegetable and seed from different accessions of Amaranthus in South Africa. Water SA 2007;33:377-80.
41. Modi AT. Growth temperature and plant age influence on nutritional quality of Amaranthus leaves and seed germination capacity. Water SA 2007;33:369-375.
42. Odhav B, Beekrum S, Akula U, Baijnat H. Preliminary assessment of nutritional value of traditional vegetables in KwaZulu-Natal, South Africa. J Food Comp Anal 2007;20:430-5.
43. Galvano F, Piva A, Ritieni A, Galvano G. Dietary strategies to counteract the effects of mycotoxins: a review. J Food Prot 2001;64:120-31.
44. Riley RT, Wang E, Schroeder JJ, Smith ER, Plattner RD, Abbas H, Yoo HS, Merrill AH Jr. Evidence for disruption of sphingolipid metabolism as a contributing factor in the toxicity and carcinogenicity of fumonisins. Nat Toxins 1996;4:3-15.
45. Wang E, Ross PF, Wilson TM, Riley RT, Merrill AH Jr. Alternation of serum sphingolibds upon dietary exposure of ponies to fumonisins, mycotoxins produced by Fusarium moniliforme. J Nutr 1992;122:1706-16.
46. Abdel-Wahhab MA, Hassan AM, Amer HA, Naguib KM. Prevention of fumonisin-induced maternal and developmental toxicity in rats by certain plant extracts. J Appl Toxicol 2004;24:469-74.
47. Yoo H, Norred WP, Wang E, Merrill AH Jr, Riley RT. Sphingosine inhibition of de novo sphingolipid biosynthesis and cytotoxicity are correlated in LLC-PK1 cells. Toxicol Appl Pharmacol 1992;114:9-15.
48. Abbas HK, Tanaka T, Shier WT. Biological activities of synthetic analogues of Alternaria alternate toxin (AAL-toxin) and fumonisin in plant and mammalian cell cultures. Phytochm 1995;40:1681-9.
49. Shier TW, Abbas HK, Mirocha CA. Toxicity of the mycotoxins fumonisin B1 and B2 and Alternaria alternate f. sp. Lycopersici toxin (AAL) in cultured mammalin cells. Mycopathologia 1991;116:97-104.
50. Singh KP, Roy D. Identification of novel breast tumor-specific mutation(s) in the q11.2 region of chromosome 17 by RAPD/AP-PCR fingerprinting. Gene 2001;269:33-43.
51. Bowditch BM, Albright DG, William JGK, Braun MJ. Use of randomly amplified polymorphic DNA markers in comparative genome studies. Meth Enzymol 1993;224:294-309.
52. Pietrasanta LI, Smith BL, MacLeod MC. A novel approach for analyzing the structure of DNA modified by benzo[a]pyrene diol epoxide at single-molecule resolution. Chem Res Toxicol 2000;13:351-5.
53. Mikhail FD, Jeanne C, Tatiana B, Koudriakova NG, Gerd PP. Quantitation and mapping of aflatoxin B1-induced DNA damage in genomic DNA using aflatoxin B1-8,9-epoxide and microsomal activation systems. Mut Res 1999;425:205-11.
54. Madden CR, Finegold MJ, Slagle BL. Altered DNA mutation spectrum in aflatoxin B1-treated transgenic mice that express the hepatitis B virus X protein. J Virol 2002;76:11770-4.
55. Lewis PD, Parry JM. In Silico p53 mutation hotspots in lung cancer. Carcinogenesis 2004;25:1099-107.
56. Jones C, Kortenkamp A. RAPD library fingerprinting of bacterial and human DNA applications in mutation detection. Carcinog Mutagen 2000;20:49-63.
57. Knasmüller S, Cavin C, Chakraborty A, Darroudi F, Majer BJ, Huber WW, Ehrlich VA. Structurally related mycotoxins ochratoxin A, ochratoxin B, and citrinin differ in their genotoxic activities and in their mode of action in human-derived liver (HepG2) cells: implications for risk assessment. Nutr Cancer 2004;2:190-7.
58. Rizzo AF, Atroshi F, Ahotupa M, Sankari S, Elovaara E. Protective effect of antioxidants against free radical-mediated lipid peroxidation induced by DON or T-2 toxin. J Vet Med Assoc 1994;41:81-90.
59. Guerra MC, Galvano F, Bonsi L, Speroni E, Costa S, Renzulli C, Cervellati R. Cyaniding-3-0-β-glucopyranoside, a natural free-radical scavenger against aflatoxin B1-and ochratoxin A-induced cell damage in a human hepatoma cell line (hep G2) and a human colonic cell line (CaCo-2). Brit J Nutr 2005;94:211-20.
60. Zeashan H, Amresh G, Singh S, Rao CV. Hepatoprotective activity of Amaranthusspinosus in experimental animals. Food Chem Toxicol 2008;46:3417-21.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Ibrahim MIM, Pieters R, Abdel-Aziem SH, Walt AM, Bezuidenhout CC, Giesy JP, Abdel-Wahhab MA. Protective effects of Amaranthus hybridus against aflatoxin B1 and fumonisin B1-induced genotoxicity in H4IIE-luc cells. Hepatoma Res 2015;1:136-46. http://dx.doi.org/10.4103/2394-5079.167377
AMA Style
Ibrahim MIM, Pieters R, Abdel-Aziem SH, Walt AM, Bezuidenhout CC, Giesy JP, Abdel-Wahhab MA. Protective effects of Amaranthus hybridus against aflatoxin B1 and fumonisin B1-induced genotoxicity in H4IIE-luc cells. Hepatoma Research. 2015; 1(-1): 136-46. http://dx.doi.org/10.4103/2394-5079.167377
Chicago/Turabian Style
Ibrahim, Mohamed I. M., Rialet Pieters, Sekena H. Abdel-Aziem, Anna M. van der Walt, Cornelius C. Bezuidenhout, John P. Giesy, Mosaad A. Abdel-Wahhab. 2015. "Protective effects of Amaranthus hybridus against aflatoxin B1 and fumonisin B1-induced genotoxicity in H4IIE-luc cells" Hepatoma Research. 1, no.-1: 136-46. http://dx.doi.org/10.4103/2394-5079.167377
ACS Style
Ibrahim, MIM.; Pieters R.; Abdel-Aziem SH.; Walt AM.; Bezuidenhout CC.; Giesy JP.; Abdel-Wahhab MA. Protective effects of Amaranthus hybridus against aflatoxin B1 and fumonisin B1-induced genotoxicity in H4IIE-luc cells. Hepatoma. Res. 2015, 1, 136-46. http://dx.doi.org/10.4103/2394-5079.167377
About This Article
Special Issue
Copyright
Data & Comments
Data
 Cite This Article 1 clicks
Cite This Article 1 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.