Interventional radiology for post-transplant anastomotic complications
Abstract
The effectiveness of percutaneous interventional radiology for anastomotic stricture in hepatic vein, portal vein, and biliary tract after living donor liver transplantation (LDLT) is described. As a number of patients with LDLT are infants < 10-year-old in the study, the first treatment option was balloon dilatation, not primary stenting. However, stent placement was performed in patients with recurrent, repeated stenosis.
Keywords
Introduction
Liver transplantation is an established treatment for end-stage liver disease[1]. The recent advances in surgical techniques and immunosuppression have led to improvements of post-transplant outcomes but various complications including bleeding, infections, rejection, vascular complications at the anastomotic site, and biliary complication will occur after liver transplantations[2,3]. Although deceased donor liver transplantation is considered a standard procedure, living donor liver transplantation (LDLT) has been widely performed owing to the shortage of donors[4]. LDLT is technically demanding because of the use of short vascular pedicles, which are more likely to cause postoperative vascular complications, such as hepatic venous outflow obstruction (HVOO) at the anastomotic site and anastomotic portal vein stenosis (PVS)[5-8]. Moreover, biliary complications remain common after LDLT, and some studies suggested that biliary stricture at the anastomotic site occurs more frequently in post-LDLT patients than in deceased liver transplantation. This is because of the small diameter of the anastomotic portion of the bile duct, anatomical diversity of the bile ducts or the complicated nature of the surgical procedure[9,10].
In this study, the effectiveness of interventional radiology (IR) for anastomotic complications after LDLT mainly in pediatric patients was described.
IR for HVOO
Vascular complications after liver transplantation include occlusion/stenosis at the site of anastomosis of hepatic artery, portal vein and hepatic vein. Although HVOO is an uncommon complication after liver transplantation, it is still an important cause of graft failures after liver transplantation[2]. The incidence of HVOO after orthotropic liver transplantation is reported to be about 1% and that after LDLT is reported to be about 2-4%[11,12]. This is because an anastomotic orifice is small and the grafts grow in LDLT. The causes of HVOO were stretching, twist and compression of hepatic vein with graft growing and adhesion change at anastomotic site[13].
HVOO are suspected with the findings of intractable ascites, abnormal venous flow patterns at Doppler ultrasonography (US), histologic findings suggesting venous congestion, or deterioration of liver function not otherwise explained. Doppler US is a useful modality for diagnosing HVOO whose findings is disappearance of pulsatile hepatic venous flow or flatness of the hepatic venous wave.
Percutaneous balloon dilatation is a safe and effective method of treating HVOO. In our study balloon dilatation is performed for patients with initial HVOO after LDLT, and expandable metallic stent placement is tried in patients with repeated HVOO after the balloon dilatation. This strategy is based on three our concepts. First, routine primary stenting may result in unnecessary placement of an expandable metallic stent. Second, long-term patency for metallic stent for decades is unknown in pediatric patients. Because infant and young patients grow, it is unknown whether their growth can match to the unchanged size of implanted expandable metallic stent. Third, implanted expandable metallic stent may disturb re-transplantation. At re-transplantation, the presence of expandable metallic stent in the wall of the suprahepatic inferior vena cava might be technically a challenge for surgeons.
Procedures
The approach to the hepatic vein is made through transjugular or transhepatic method. After passage of the catheter through the stenotic segment of the hepatic vein, venography and manometry; measurement of venous pressure of proximal and distal sides of the stenosis and the pressure gradient across the stricture is performed. Patients with a pressure gradient of more than 3 mmHg are considered to have significant outflow obstruction and are candidates for balloon dilatation.
Balloon dilatation [Figure 1] is performed following venography with a 7.0-Fr percutaneous transluminal angioplasty catheter with a balloon diameter of 6-12 mm. The balloon is inflated three times for 60 s with an atmospheric pressure of 10 atm. The diameter of the balloon is the same as the vein on the mesenteric side of the stenosis. The balloon is routinely inflated 3 times for 60 s with an atmospheric pressure of 10 atm. In patients showing recurrent HVOO, the stent placement [Figure 2] is performed. We used a self-expanding metallic stent with a diameter 20-30% larger than that of the hepatic vein.
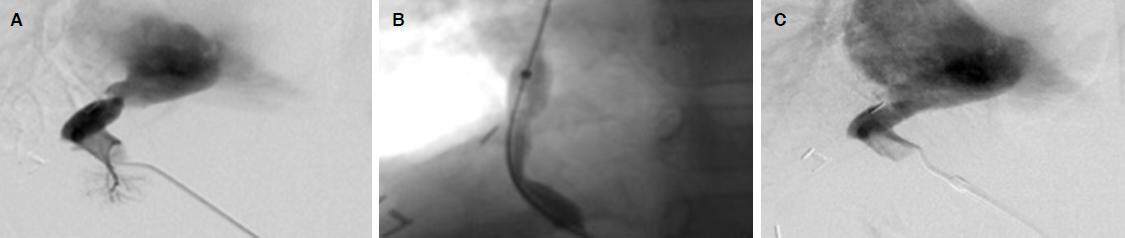
Figure 1. A 6-year-old boy with biliary atresia underwent left-lobe LDLT, HVOO was diagnosed 5.1 years after LDLT, and hepatic venography was performed. (A) preoperative venogram showing an anastomotic stricture. As to the manometry finding, the pressure gradient, HV-RA was 12 mmHg; (B) fluoroscopic view during balloon dilatation showing the notch of the balloon at the stenosis; (C) preoperative venogram after the balloon dilatation showing improvement of the stenosis. The pressure gradient improved; HV-RA was 2 mmHg. LDLT: living donor liver transplantation; HVOO: hepatic venous outflow obstruction; HV: hepatic vein; RA: right atrium
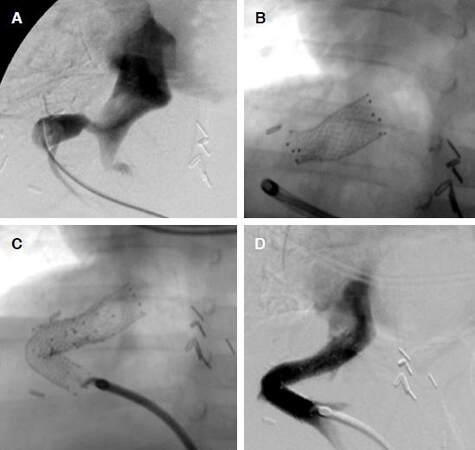
Figure 2. A 1-year-old girl with biliary atresia underwent left-lobe LDLT, HVOO repeated after 3-sessions of balloon dilatation, and stent placement was performed. (A) preoperative hepatic venogram showing an anastomotic stricture; (B) fluoroscopic view after stent placement. However, HVOO repeated, and additional stent placement was performed twice. After the 3rd stent placement, HV was patent, and no HVOO was noted for 5 years. (C) fluoroscopic view the 3rd after stent placement; (D) hepatic venogram showing no anastomotic stricture. LDLT: living donor liver transplantation; HVOO: hepatic venous outflow obstruction
Results
In our reported study[14], the rates of technical success, primary patency and primary-assisted patency were evaluated. Technical success is defined as success in interventional procedures. Primary patency is defined as the interval between the initial balloon angioplasty and recurrent HVOO necessitating percutaneous intervention. Primary-assisted patency is defined as patency following the initial angioplasty until repeated percutaneous intervention therapy is discontinued.
We performed IR for 48 patients with HVOO after LDLT whose follow-up periods ranged from 1 to 182 months (median, 51.5 months). Technical success was achieved in 92 of 93 sessions (99%) and in 47 of 48 patients (98%). The primary and primary assisted patency at 1, 3, 5, 10 years after the initial privacy threshold analysis (PTA) were 64%, 57%, 57%, 52% and 98%, 95%, 95%, and 95% respectively.
IR for PVS
The rate of PV complications after deceased donor liver transplantation has been reported to be < 3%[7]. However, in patients with reduced-size liver transplantation or LDLT, the rate of PV complication can be higher (9-14%) than in patients with conventional deceased donor liver transplantation[7,15]. PV complications are divided mainly into anastomotic PVS and portal vein thrombosis (PVT)[16]. Anastomotic PVS can lead to graft failure if not properly treated. The treatment options for PVS after liver transplantation are surgical treatment and percutaneous interventions, including percutaneous balloon dilatation and stent placement. However, surgical treatment of these complications has been limited owing to technical difficulties or invasiveness. Currently, the surgical treatment of PVS after liver transplantation has been replaced by percutaneous balloon dilatation and stent placement, because of lower invasiveness and greater effectiveness.
PVS was clinically suspected with the following findings: (1) clinical symptoms of portal hypertension, such as ascites, splenomegaly, gastrointestinal tract bleeding from varices, and thrombocytopenia; and (2) US findings, including greater than 50% stenosis (the diameter of stenosis/the diameter of a main PV on the mesenteric side) or no flow in the PV; or the presence of an acceleration of flow at the stenosis or a post-stenotic jet flow or minimal flow in the intrahepatic PV on Doppler US. Our inclusion criteria for PVS were: (1) greater than 50% stenosis (the diameter of the stenosis/the diameter of a PV on the distal side); or (2) > 5 mmHg pressure gradient across the stenosis between the proximal and distal PV.
Procedures
The approach to the intrahepatic PV is transhepatic at the first session of percutaneous intervention. Balloon dilatation [Figure 3] is performed following portography with a 7.0-Fr percutaneous transluminal angioplasty catheter with a balloon diameter of 6-12 mm. The balloon is inflated three times for 60 s with an atmospheric pressure of 10 atm. The diameter of the balloon is the same as the vein on the mesenteric side of the stenosis. The balloon is routinely inflated three times for 60 s with an atmospheric pressure of 10 atm. Stent placement [Figure 4] is performed in patients who developed recurrent PVS. We used a self-expanding metallic stent with a diameter 20-30% larger than that of the PV proximal to the stenosis and with sufficient length to cover the stricture. In patients where the percutaneous transhepatic approach to the PV is unsuccessful, or where placing a metallic stent with the percutaneous transhepatic approach might be technically difficult owing to a severely curved PV, a transileocecal approach is chosen following laparotomy.
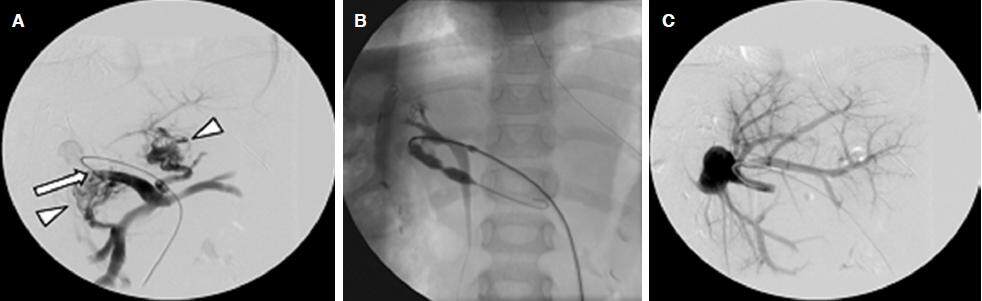
Figure 3. A 7-year-old girl with biliary atresia underwent left-lobe LDLT, PVS was suspected 5 years after LDLT, and portography was performed. (A) pretreatment portogram showing an anastomotic stricture (arrow), collateral vessels (arrowhead), and poor flow through the intrahepatic portal vein; (B) fluoroscopic view during balloon angioplasty showing the notch of the balloon at the stenosis; (C) portogram after the balloon angioplasty showing improved blood flow through the portal vein and disappearance of collateral vessels. PVS did not recur after the balloon angioplasty; LDLT: living donor liver transplantation; PVS: portal venous stenosis
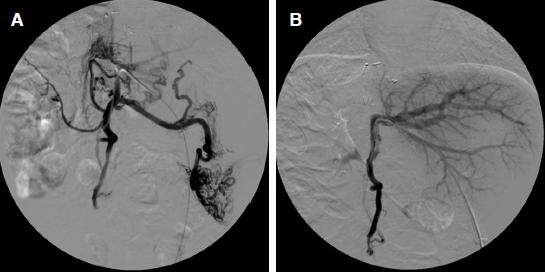
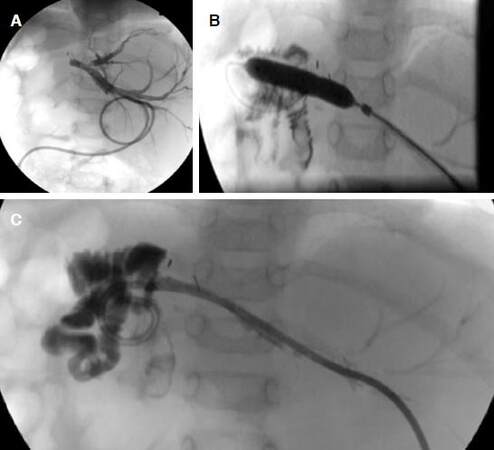
Figure 4. A 2-year-old girl with biliary atresia had undergone left-lobe LDLT and seven sessions of balloon angioplasty for PVS, because recurrent PVS was suspected, portography was performed. (A) pretreatment portogram showing a severe anastomotic stricture and no flow into the intrahepatic portal vein; (B) portogram after stent placement showing improved blood flow into the portal vein, PVS did not recur after stent placement; LDLT: living donor liver transplantation; PVS: portal venous stenosis
Results
In our reported study[17], the rates of technical success, primary patency and primary-assisted patency were evaluated. Technical success is defined as success in interventional procedures. Primary patency is defined as the interval between the initial balloon angioplasty and recurrent PVS necessitating percutaneous intervention. Primary-assisted patency is defined as patency following the initial angioplasty until repeated percutaneous intervention therapy is discontinued.
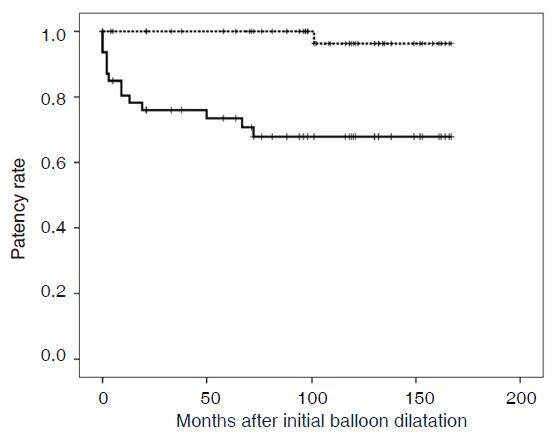
We performed IR for the 43 patients with PVS after LDLT, whose follow-up periods ranged from 5 to 169 months (mean, 119 months). Technical success was achieved in 65 of 66 sessions (98%) and in 42 of 43 patients (98%). The primary and primary assisted patency at 1, 3, 5, 10 years after the initial PTA were 83%, 78%, 76%, and 70%, respectively, and 100%, 100%, 100%, and 96%, respectively [Figure 5].
Figure 5. Kaplan-Meier curve showing primary- and primary-assisted patency rates. Solid and dotted lines indicate primary patency and primary-assisted patency, respectively. Vertical lines on both lines indicate censored observations. At 1, 3, 5, and 10 years after the first balloon angioplasty the primary patency rates were 80%, 76%, 73%, and 67%, respectively, and the primary-assisted patency rates were 100%, 100%, 100%, and 96% respectively
IR for anastomotic biliary stenosis
Anastomotic biliary stricture is the most common biliary complication. Some studies have suggested that biliary stricture occurs more frequently in post-LDLT patients than in deceased liver transplantation because of the small diameter of the anastomotic portion of the bile duct, anatomical diversity of the bile ducts, or the complicated nature of the surgical procedure[9,10,18]. There are two strategies for treating anastomotic strictures: via the endoscopic retrograde approach[19] or the percutaneous transhepatic approach[20]. The endoscopic retrograde approach is feasible for post-transplant patients with a duct-to-duct anastomosis, and endoscopic stent placement has been reported to be effective for biliary strictures in post-transplant patients[21]. Because the most common disease in pediatric patients with LDLT has been biliary atresia, most of them have undergone Kasai’s surgery and Roux-en-Y hepaticojejunostomy (RYHJ). Thus, percutaneous transhepatic biliary drainage (PTBD) is believed to be a first-line treatment for biliary strictures in pediatric patients who underwent LDLT with RYHJ.
An anastomotic biliary stricture is suspected based on laboratory, US, cholescintigraphic findings, and liver biopsy results. Liver function tests show increases in total bilirubin, direct bilirubin, aspartate aminotransferase (AST), alanine aminotransferase (ALT), r-glutamyl transpeptidase (r-GTP), and/or alkaline phosphatase (ALP). US findings that suggest anastomotic stricture are dilatation of intrahepatic bile ducts that appeared during the follow-up. Cholescintigraphy shows delayed visualization of the bowel (> 10 min after injection of the radiotracer (99mTc-N-pyridoxyl-5-methyltryptophan). Liver biopsy reveals cholestasis.
Procedures
Access to the biliary duct was made under US guidance. After puncture of a biliary duct with a 21-gauge needle under US guidance and opacifying the biliary duct (percutaneous transhepatic cholangiography), PTBD is performed using a 0.018-inch guidewire and a 5-Fr catheter [Figure 6]. Then, passage through the anastomotic biliary stricture is attempted with a 0.035-inch hydrophilic guidewire and a 5-Fr catheter. After successful passage of the catheter and exchange of a 7-Fr interventional sheath introducer [Figure 6], dilatation was performed with a balloon catheter (diameter; 4-10 mm). The diameter of the balloon was matched to the diameter of the intrahepatic bile duct on the hepatic side of stricture. The balloon was placed across the stricture and inflated for 180 s with an atmospheric pressure of 10 atm. After balloon dilatation, cholangiography was repeated to evaluate the effectiveness. Then, an 8.5-Fr internal-external drainage tube (Pig-tail catheter, Cook; IN, USA) was placed, covering the Roux-Y jejunum and intrahepatic bile ducts across the anastomotic stricture.
Figure 6. A 2-year-old boy who had undergone LDLT 20 months ago was suspected of having a biliary anastomotic stricture. (A) PTC showing an anastomotic stricture; (B) fluoroscopic view during balloon dilatation. Balloon dilatation was performed at 10 atm for 3 min using a 6-mm-diameter balloon catheter; (C) fluoroscopic view shows an 8.5-Fr. internal-external drainage tube placed across the anastomotic stricture. After serial exchange with a larger diameter catheter (16-Fr), the drainage tube was removed. No recurrent stricture was noted for 117 months after the biliary interventions. LDLT: living donor liver transplantation; PTC: percutaneous transhepatic cholangiography
Serial exchanges for a larger 14-Fr or 16-Fr drainage tube with or without balloon dilations were routinely performed at 1- to 6-week intervals. At a follow-up session, cholangiography was performed to evaluate the persistence of stricture. If the stricture had widened and the laboratory data had resolved, the tube was removed.
Results
In our reported study[22], clinical success, tube independent rate, and patency rate were evaluated. Clinical success is defined as resolution or marked improvement of clinical symptoms including fever, and improvement of laboratory findings, including the serum levels of AST, ALT, total bilirubin, direct bilirubin, r-GTP, and ALP. Tube independent rate is defined as the rate at which the patient can undergo tube removal after symptoms are diminished and laboratory findings have improved. Patency rate is estimated by the Kaplan-Meier analysis. Primary patency is defined as the interval between placement of an internal drainage tube and appearance of a recurrent biliary stricture necessitating percutaneous biliary interventions. Primary-assisted patency is defined as the interval between placement of an internal drainage tube and when treatment with repeated percutaneous interventions is discontinued.
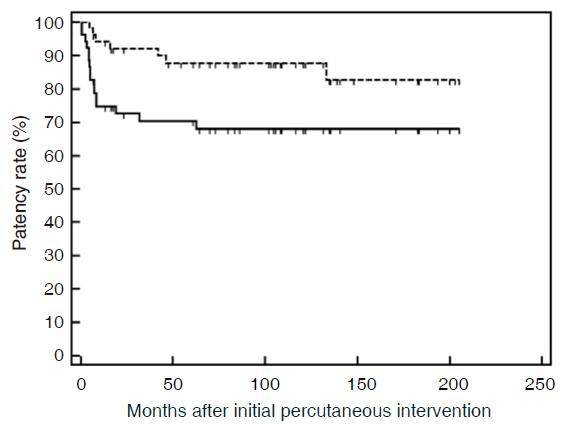
We performed IR for the 52 patients with anastomotic biliary stenosis after LDLT, whose follow-up periods ranged from 5 to 206 months (median, 100 months). Clinical success was noted in 43 of 52 patients (83%). Removal of the drainage tube was achieved in 49 of 52 patients (94%). Of the three patients having a drainage tube, two underwent surgical reanastomosis, and one had a drainage tube implanted subcutaneously. The primary patency rates at 1, 3, 5, and 10 years after the initial drainage tube placement were 75%, 70%, 70%, and 68%, respectively. The primary-assisted patency rates at 1, 3, 5, and 10 years after the initial drainage tube placement were 94%, 92%, 88%, and 88%, respectively [Figure 7].
Figure 7. Kaplan-Meier curve showing primary- and primary-assisted patency rates. Solid and dotted lines indicate primary patency and primary-assisted patency, respectively. Vertical lines on both lines indicate censored observations. The primary patency rates at 1, 3, 5, and 10 years after the initial drainage tube placement were 75%, 70%, 70%, and 68%, respectively. The primary-assisted patency rates at 1, 3, 5, and 10 years after the initial drainage tube placement were 94%, 92%, 88%, 88%, respectively
Conclusion
In conclusion, percutaneous IR is a minimally invasive, effective treatment for HVOO, PVS, and anastomotic biliary stricture after LDLT.
Declarations
Authors’ contributionsThe author contributed solely to this paper.
Financial support and sponsorshipNone.
Conflicts of interestThere are no conflicts of interest.
Patient consentNot applicable.
Ethics approvalThis review paper is waived for ethics approval.
Copyright© The Author(s) 2017.
REFERENCES
2. Buell JF, Funaki B, Cronin DC, Yoshida A, Perlman MK, Lorenz J, Kelly S, Brady L, Leef JA, Millis JM. Long-term venous complications after full-size and segmental pediatric liver transplantation. Ann Surg 2002;236:658-66.
3. Patkowski W, Nyckowski P, Zieniewicz K, Pawlak J, Michalowicz B, Kotulski M, Smoter P, Grodzicki M, Skwarek A, Ziolkowski J, Oldakowska-Jedynak U, Niewczas M, Paczek L, Krawczyk M. Biliary tract complications following liver transplantation. Transplant Proc 2003;35:2316-7.
4. Tanaka K, Uemoto S, Tokunaga Y, Fujita S, Sano K, Nishizawa T, Sawada H, Shirahase I, Kim HJ, Yamaoka Y. Surgical techniques and innovations in living related liver transplantation. Ann Surg 1993;217:82-91.
5. Egawa H, Inomata Y, Uemoto S, Asonuma K, Kiuchi T, Okajima H, Yamaoka Y, Tanaka K. Hepatic vein reconstruction in 152 living-related donor liver transplantation patients. Surgery 1997;121:250-7.
6. Settmacher U, Nüssler NC, Glanemann M, Haase R, Heise M, Bechstein WO, Neuhaus P. Venous complications after orthotopic liver transplantation. Clin Transplant 2000;14:235-41.
7. Moon JI, Jung GO, Choi GS, Kim JM, Shin M, Kim EY, Kwon CH, Kim SJ, Joh JW, Lee SK. Risk factors for portal vein complications after pediatric living donor liver transplantation with left-sided grafts. Transplant Proc 2010;42:871-5.
8. Darwish AA, Bourdeaux C, Kader HA, Janssen M, Sokal E, Lerut J, Ciccarelli O, Veyckemans F, Otte JB, de Goyet Jde V, Reding R. Pediatric liver transplantation using left hepatic segments from living related donors: surgical experience in 100 recipients at Saint-Luc University Clinics. Pediatr Transplant 2006;10:345-53.
9. Busuttil RW, Farmer DG, Yersiz H, Hiatt JR, McDiarmid SV, Goldstein LI, Saab S, Han S, Durazo F, Weaver M, Cao C, Chen T, Lipshutz GS, Holt C, Gordon S, Gornbein J, Amersi F, Ghobrial RM. Analysis of long-term outcomes of 3200 liver transplantations over two decades: a single-center experience. Ann Surg 2005;241:905-16; discussion 916-8.
10. Seo JK, Ryu JK, Lee SH, Park JK, Yang KY, Kim YT, Yoon YB, Lee HW, Yi NJ, Suh KS. Endoscopic treatment for biliary stricture after adult living donor liver transplantation. Liver Transpl 2009;15:369-80.
11. Chen HL, Concejero AM, Huang TL, Chen TY, Tsang LL, Wang CC, Wang SH, Chen CL, Cheng YF. Diagnosis and interventional radiological treatment of vascular and biliary complications after liver transplantation in children with biliary atresia. Transplant Proc 2008;40:2534-6.
12. Ko GY, Sung KB, Yoon HK, Kim JH, Song HY, Seo TS, Lee SG. Endovascular treatment of hepatic venous outflow obstruction after living-donor liver transplantation. J Vasc Interv Radiol 2002;13:591-9.
13. Carnevale FC, Machado AT, Moreira AM, De Gregorio MA, Suzuki L, Tannuri U, Gibelli N, Maksoud JG, Cerri GG. Midterm and long-term results of percutaneous endovascular treatment of venous outflow obstruction after pediatric liver transplantation. J Vasc Interv Radiol 2008;19:1439-48.
14. Yabuta M, Shibata T, Shibata T, Shinozuka K, Isoda H, Okamoto S, Uemoto S, Togashi K. Long-term outcome of percutaneous interventions for hepatic venous outflow obstruction after pediatric living donor liver transplantation: experience from a single institute. J Vasc Interv Radiol 2013;24:1673-81.
15. Woo DH, Laberge JM, Gordon RL, Wilson MW, Kerlan RK, Jr. Management of portal venous complications after liver transplantation. Tech Vasc Interv Radiol 2007;10:233-9.
16. Olcott EW, Ring EJ, Roberts JP, Ascher NL, Lake JR, Gordon RL. Percutaneous transhepatic portal vein angioplasty and stent placement after liver transplantation: early experience. J Vasc Interv Radiol 1990;1:17-22.
17. Yabuta M, Shibata T, Shibata T, Shinozuka K, Isoda H, Okamoto S, Uemoto S, Togashi K. Long-term outcome of percutaneous transhepatic balloon angioplasty for portal vein stenosis after pediatric living donor liver transplantation: a single institute's experience. J Vasc Interv Radiol 2014;25:1406-12.
18. Trotter JF, Wachs M, Everson GT, Kam I. Adult-to-adult transplantation of the right hepatic lobe from a living donor. N Engl J Med 2002;346:1074-82.
19. Yazumi S, Yoshimoto T, Hisatsune H, Hasegawa K, Kida M, Tada S, Uenoyama Y, Yamauchi J, Shio S, Kasahara M, Ogawa K, Egawa H, Tanaka K, Chiba T. Endoscopic treatment of biliary complications after right-lobe living-donor liver transplantation with duct-to-duct biliary anastomosis. J Hepatobiliary Pancreat Surg 2006;13:502-10.
20. Zajko AB, Sheng R, Zetti GM, Madariaga JR, Bron KM. Transhepatic balloon dilation of biliary strictures in liver transplant patients: a 10-year experience. J Vasc Interv Radiol 1995;6:79-83.
21. Kurita A, Kodama Y, Minami R, Sakuma Y, Kuriyama K, Tanabe W, Ohta Y, Maruno T, Shiokawa M, Sawai Y, Uza N, Yazumi S, Yoshizawa A, Uemoto S, Chiba T. Endoscopic stent placement above the intact sphincter of Oddi for biliary strictures after living donor liver transplantation. J Gastroenterol 2013;48:1097-104.
22. Imamine R, Shibata T, Yabuta M, Shinozuka K, Kataoka M, Isoda H, Yoshizawa A, Uemoto S, Togashi K. Long-term outcome of percutaneous biliary interventions for biliary anastomotic stricture in pediatric patients after living donor liver transplantation with Roux-en-Y hepaticojejunostomy. J Vasc Interv Radiol 2015;26:1852-9.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Shibata T. Interventional radiology for post-transplant anastomotic complications. Hepatoma Res 2017;3:221-7. http://dx.doi.org/10.20517/2394-5079.2017.34
AMA Style
Shibata T. Interventional radiology for post-transplant anastomotic complications. Hepatoma Research. 2017; 3: 221-7. http://dx.doi.org/10.20517/2394-5079.2017.34
Chicago/Turabian Style
Shibata, Toshiya. 2017. "Interventional radiology for post-transplant anastomotic complications" Hepatoma Research. 3: 221-7. http://dx.doi.org/10.20517/2394-5079.2017.34
ACS Style
Shibata, T. Interventional radiology for post-transplant anastomotic complications. Hepatoma. Res. 2017, 3, 221-7. http://dx.doi.org/10.20517/2394-5079.2017.34
About This Article
Copyright
Data & Comments
Data
 Cite This Article 3 clicks
Cite This Article 3 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.