Hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in sub-Saharan Africa: no room for complacency
The increasing incidence and mortality of hepatocellular carcinoma (HCC) driven by hepatitis B virus (HBV) infection is a global public health concern[1]. Despite World Health Organization (WHO) targets for the elimination of hepatitis as a public health problem by 2030, death attributable to viral hepatitis has continued to rise over the past decade[2], in contrast to the reduction seen in the comparable threats of human immunodeficiency virus (HIV), tuberculosis (TB) and malaria.
Africa accounts for 17% of the global population[3], but a disproportionate 20%-30% of all those living with HBV infection reside in sub-Saharan Africa (sSA)[4]. Likewise, while HCC is the 6th commonest cancer worldwide, in parts of Western and Central Africa, it is the highest cause of cancer mortality in men and the third in women[5], primarily accounted for by HBV infection[6]. In sSA, the recorded annual death from HBV-associated HCC is virtually the same as the total number of HCC cases diagnosed in the population. This high annual fatality ratio of close to 1 results from the characteristic delayed presentation, aggressive clinical course, and paucity of treatment options, and highlights inadequacies in prevention, screening, and treatment programs. Furthermore, HCC may occur in young adults, in the absence of underlying cirrhosis, and in the context of low viral loads and HBeAg-negativity and occult HBV, making targeted surveillance difficult. The burden of disease is complicated and enhanced by overlap with exposure to other carcinogenic influences, including aflatoxin, alcohol, HCV infection, and fatty liver disease[5,7].
The “Find the Missing Millions” campaign spearheaded by the World Hepatitis Alliance[8] highlights the need for improved diagnosis of HBV infection. Through linkage to clinical care, individuals can then be treated with antivirals and monitored to reduce the risk of evolution and progression of HCC. To support these programmes, there is a need to develop and decentralise infrastructure and human resources for HBV, and link activities to local and regional activities already successfully established for other infections, including HIV, TB and malaria[9]. There is a particularly strong rationale for the alignment of HBV with HIV services and infrastructure to support programmes both for HBV monoinfection and for patients with dual infection. Ultimately, elimination of HBV will require a substantial scale-up of diagnosis and treatment, with advances in treatment that deliver functional cure rather than just virologic suppression[9]. For this to be a realistic prospect in Africa, platforms for clinical trials need to be developed, with clinical cohorts that are well characterised, linked to consistent healthcare services, and established on standard-of-care treatment.
Activities for HBV networks must also focus on interrupting HBV transmission. This includes enhanced coverage with three-dose vaccine coverage, with the first dose given at birth in endemic settings, alongside other interventions to prevent mother-to-child transmission (PMTCT), screening to prevent transfusion-transmitted infections, and harm reduction programmes to prevent transmission in people-who-inject-drugs.
For many populations in Africa, the epidemiology, aetiology and characteristics of liver disease are not well characterised. Settings across sSA are highly heterogeneous in their population characteristics, health challenges and priorities, resources, partnerships and infrastructure. For this reason, local investment and enterprise are fundamental to driving interventions that best fit the local populations, while also recognising the need for international collaborations that facilitate sharing of knowledge, resources, and experience. There remain significant gaps in our understanding of the biology and underlying mechanisms of pathogenesis in HBV infection, especially with the diverse (sub)genotypes prevailing in Africa. There is an urgent need to better understand the molecular basis of diverse outcomes, including cirrhosis, cancer and occult infection. To tackle HBV and HCC, interdisciplinary action is needed to build partnerships between clinical domains of oncology, hepatology, infectious diseases and public health, combined with social sciences, health economics and policy development. Approaches to surveillance and risk-stratification must be enhanced through the development and validation of new biomarkers, better access to imaging, and development of risk scores, with attention to the performance, accessibility, scalability and cost of these approaches in different settings.
The collection of reliable open-access cancer data representing African populations will be an important driver of progress[10]. There is promise in the establishment of cancer registries such as local and national enterprises (for example, a cancer registry in Nigeria[11]) and the wider African Cancer Registry Network, which had grown to represent 25 countries by 2020[12,13]. However, progress has been slow, and thwarted by a lack of investment and infrastructure (with particular challenges for rural regions). Gaps in data are substantial: only a minority of the population of sSA is covered by accurate death registration systems (estimated as 0.25% of the population in 2005[14] and 10% by 2015[13]), and only 11 countries have a national cancer control plan[15]. The scale-up of biobanks and cancer registries will lead to enhanced data to underpin risk assessment, inform clinical practice, and drive appropriate research in different population settings, ultimately underpinning policy and resource allocation. This requires significant investment in human resources and infrastructure.
In sSA, stigma and discrimination are substantial challenges for HBV, compounded by poor education and awareness, and lack of role models[16,17]; there is an urgent need to enhance knowledge and to promote the patient voice[18]. Mobilising civil society is also key to increasing awareness and advocacy, and improving participation in research with a patient-centric focus. Raising the profile of HBV infection and its associated complications will help tackle political inertia in combating it, increasing advocacy to harness the required resources, and building and sustaining interventions.
The Covid-19 pandemic (caused by SARS-CoV-2) has amplified many challenges that are pertinent to HBV control, including HBV vaccination, disrupting existing healthcare infrastructure, diverting and depleting resources, and highlighting striking inequities in vaccine distribution and roll out[19]. However, as accelerated efforts to combat the pandemic drive establishment of vaccine production plants in Africa, there is also much to be gained from innovation, infrastructure and investment[20]. African initiatives to address SARS-CoV-2 have demonstrated the opportunities, reach and impact of locally driven enterprise[21], and the continent must continue to build on this success and momentum. The pace of progress to tackle Covid-19 raises questions as to why this field has been so neglected to date, and why the modest funding for birth dose HBV vaccination has not been made consistently available. HBV has always been overshadowed by the infection “blockbusters” AIDS, TB, malaria, and now Covid-19.
The pandemic highlights how Africa’s challenges can only be addressed through a combination of efforts at local and regional levels, together with sustained investment from the global community, with international and intercontinental partnerships to support the deployment of resources and skills and transparent sharing of data in the public domain. The establishment of the African CDC, launched in January 2017, is an important step[22]. Decolonisation of global health is a priority for Africa, which has long suffered “parachute research” whereby clinical and research teams from high-income countries form superficial or temporary relationships with their counterparts in low/middle-income settings to access samples and data[23,24]. Formal pathways to equitable collaboration are being established[25], but much work is still needed in this domain.
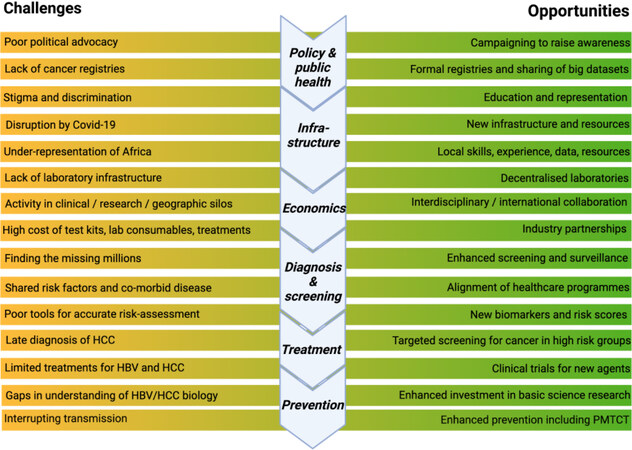
Building sustainable progress to tackle the burden of HBV and HCC requires a concerted effort to sensitise policy and decision makers to the challenges faced in sSA. There is increasing awareness and despite major challenges remaining on the road to elimination[26], there are many opportunities for intervention [Figure 1] through enhanced local action, international partnerships, and investment in education, clinical services, basic and translational research and public health practice to accelerate progress towards elimination targets.
Figure 1. Challenges and opportunities for the elimination of hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in Africa. Figure created using Biorender.com with license to publish. PMTCT: Prevention of mother to child transmission.
DECLARATIONS
Authors’ contributionsWrote and reviewed the article: Matthews PC, Kramvis A
Availability of data and materialsNot applicable.
Financial support and sponsorshipPhilippa Matthews receives funding from the Wellcome Trust (Grant ref 110110/Z/15/Z) and from the Francis Crick Institute, London.
Conflicts of interestPhilippa Matthews supervises a PhD student who is co-funded by GSK. Kramvis A declared that there are no conflicts of interest.
Ethical approval and consent to participateNot applicable.
Consent for publicationNot applicable.
Copyright© The Author(s) 2022.
REFERENCES
1. World Health Organization Fact Sheet: Hepatitis B Virus. Available from: https://www.who.int/news-room/fact-sheets/detail/hepatitis-b [Last accessed on 24 Mar 2022].
2. Global health sector strategy on viral hepatitis 2016-2021. Towards ending viral hepatitis. Available from: https://apps.who.int/iris/handle/10665/246177 [Last accessed on 24 Mar 2022].
3. Distribution of the global population 2021, by continent. Available from: https://www.statista.com/statistics/237584/distribution-of-the-world-population-by-continent/ [Last accessed on 24 Mar 2022].
4. WHO Regional Office for Africa: Hepatitis. Available from: https://www.afro.who.int/health-topics/hepatitis [Last accessed on 24 Mar 2022].
5. Hainaut P, Amadou A, Gormally E. Cancer prevention and control: hepatocellular carcinoma. Ecancermedicalscience 2019;13:949.
6. Chen WC, Singh E, Muchengeti M, et al. Johannesburg Cancer Study (JCS): contribution to knowledge and opportunities arising from 20 years of data collection in an African setting. Cancer Epidemiol 2020;65:101701.
7. Konyn P, Ahmed A, Kim D. Current epidemiology in hepatocellular carcinoma. Expert Rev Gastroenterol Hepatol 2021;15:1295-307.
8. World Hepatitis Alliance. Find the Missing Millions Campaign. Available from: https://www.worldhepatitisalliance.org/missing-millions/about/ [Last accessed on 24 Mar 2022].
9. Kramvis A. Challenges for hepatitis B virus cure in resource-limited settings in sub-Saharan Africa. Curr Opin HIV AIDS 2020;15:185-92.
10. Omonisi AE, Liu B, Parkin DM. Population-based cancer registration in sub-Saharan Africa: its role in research and cancer control. JCO Glob Oncol 2020;6:1721-8.
11. Jedy-Agba EE, Oga EA, Odutola M, et al. Developing National Cancer Registration in Developing Countries - Case Study of the Nigerian National System of Cancer Registries. Front Public Health 2015;3:186.
12. African Cancer Registry. Available from: https://afcrn.org/index.php [Last accessed on 24 Mar 2022].
13. Gakunga R, Parkin DM. African Cancer Registry Network. Cancer registries in Africa 2014: a survey of operational features and uses in cancer control planning. Int J Cancer 2015;137:2045-52.
14. Mathers CD, Fat DM, Inoue M, Rao C, Lopez AD. Counting the dead and what they died from: an assessment of the global status of cause of death data. Bull World Health Organ 2005;83:171-7.
15. Duncan K, Cira MK, Barango P, Trimble EL. Challenges and opportunities in the creation and implementation of cancer-control plans in Africa. Ecancermedicalscience 2019;13:938.
16. O’Hara GA, McNaughton AL, Maponga T, et al. Hepatitis B virus infection as a neglected tropical disease. PLoS Negl Trop Dis 2017;11:e0005842.
18. Matthews PC, Jack K, Wang S, et al. A call for advocacy and patient voice to eliminate hepatitis B virus infection. Lancet Gastroenterol Hepatol 2022;7:282-5.
19. Pley CM, McNaughton AL, Matthews PC, Lourenço J. The global impact of the COVID-19 pandemic on the prevention, diagnosis and treatment of hepatitis B virus (HBV) infection. BMJ Glob Health 2021;6:e004275.
20. African Vaccine Manufacturing Initiative. Available from: https://www.avmi-africa.org/ [Last accessed on 24 Mar 2022].
21. Tegally H, Wilkinson E, Giovanetti M, et al. Detection of a SARS-CoV-2 variant of concern in South Africa. Nature 2021;592:438-43.
22. Africa CDC. Centers for Disease Control and Prevention. Available from: https://africacdc.org/ [Last accessed on 24 Mar 2022].
23. Heinz E, Holt KE, Meehan CJ, Sheppard SK. Addressing parachute research and removing barriers for LMIC researchers in Microbial Genomics. Microb Genom 2021;7:000722.
24. Morton B, Vercueil A, Masekela R, et al. Consensus statement on measures to promote equitable authorship in the publication of research from international partnerships. Anaesthesia 2022;77:264-76.
25. The Official PLOS Blog. Announcing a new PLOS policy on inclusion in global research. Available from: https://theplosblog.plos.org/2021/09/announcing-a-new-plos-policy-on-inclusion-in-global-research/ [Last accessed on 24 Mar 2022].
26. Maponga TG, Nwankwo C, Matthews PC. Sustainable Development Goals for HBV elimination in South Africa: challenges, progress, and the road ahead. South African Gastroenterology Review 2019;17:15-25.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Matthews PC, Kramvis A. Hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in sub-Saharan Africa: no room for complacency. Hepatoma Res 2022;8:14. http://dx.doi.org/10.20517/2394-5079.2022.11
AMA Style
Matthews PC, Kramvis A. Hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in sub-Saharan Africa: no room for complacency. Hepatoma Research. 2022; 8: 14. http://dx.doi.org/10.20517/2394-5079.2022.11
Chicago/Turabian Style
Matthews, Philippa C., Anna Kramvis. 2022. "Hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in sub-Saharan Africa: no room for complacency" Hepatoma Research. 8: 14. http://dx.doi.org/10.20517/2394-5079.2022.11
ACS Style
Matthews, PC.; Kramvis A. Hepatitis B virus (HBV) and hepatocellular carcinoma (HCC) in sub-Saharan Africa: no room for complacency. Hepatoma. Res. 2022, 8, 14. http://dx.doi.org/10.20517/2394-5079.2022.11
About This Article
Copyright
Data & Comments
Data

 Cite This Article 10 clicks
Cite This Article 10 clicks











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.