First case report of inflammatory myofibroblastic tumor of peritoneal cavity in a living donor liver transplantation recipient
Abstract
Post-transplantation malignancies are well known complications after liver transplantation. Certain malignancies are more common in pediatric recipients than adults. Inflammatory myofibroblastic tumors (IMTs) are reactive neoplasms with miniscule malignant potential. IMTs are more common after hematopoietic stem cell transplantation. However, there is 1 case reported in the literature after deceased donor liver transplantation. The authors describe a case of IMT after living donor liver transplantation. The patient was a 1-year-old girl who underwent living donor liver transplantation (LDLT) for decompensated cirrhosis secondary to extra hepatic biliary atresia. Six months post LDLT routine ultrasonography revealed multiple solid abdominal masses. Repeated biopsies were inconclusive. Hence surgical excision was carried out. Histopathological examination revealed IMT. Immunohistochemistry was positive for anaplastic-lymphoma kinase activity. Ceritinib, a tyrosine kinase inhibitor, was used as adjuvant chemotherapy for 1 year. At 1.5 years (at the time of writing this paper) of follow-up, the child was disease free on imaging (whole body positron emission tomography-computed tomography). This will be the first case of IMT after LDLT to be reported in the literature.
Keywords
Introduction
Post-transplantation malignancies are well known complication of chronic immunosuppression. Certain malignancies are more common in pediatric recipients than in adults. Literature review revealed that inflammatory myofibroblastic tumors (IMTs) generally occur 3 to 6 months post-transplantation and are more common after hematopoietic stem cell transplantation (HSCT). Epstein-Barr virus has been held responsible in some reported cases. Lungs are the commonest site however they can present anywhere in the body. We describe a case of 6 months old female child who was suffering from end stage liver disease secondary to extra-hepatic biliary atresia (EHBA). She underwent living donor liver transplantation (LDLT). Six months post liver transplantation she was found to have intra-abdominal tumors on routine ultrasonography (USG). She underwent surgical excision of peritoneal neoplasms. Histopathological examination (HPE) confirmed IMT with anaplastic lymphoma kinase activity. She received ceritinibas chemotherapy for 1 year. At 1.5 years of follow-up, the child is recurrence free.
Case report
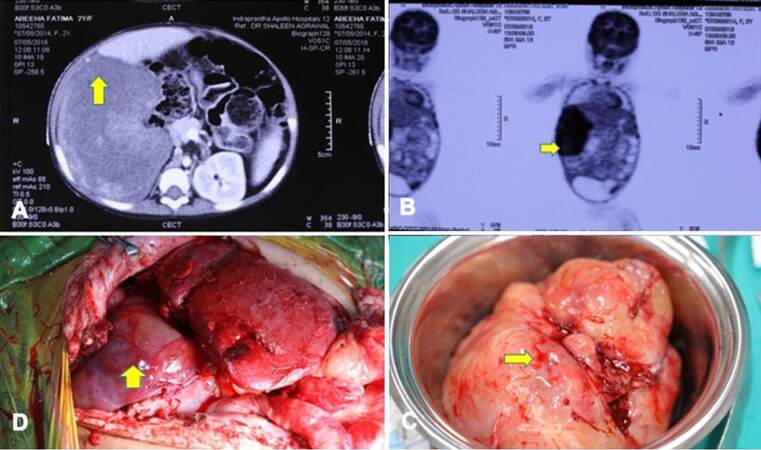
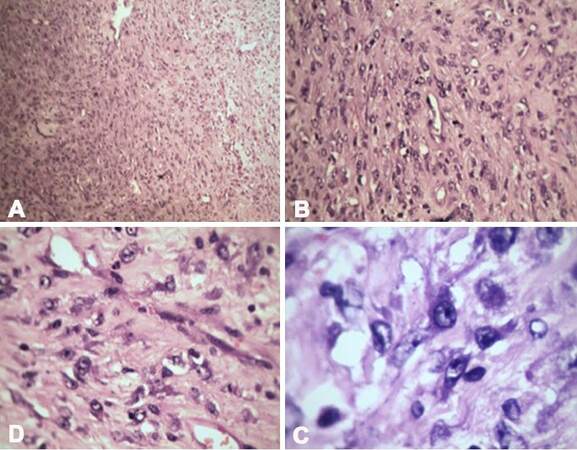
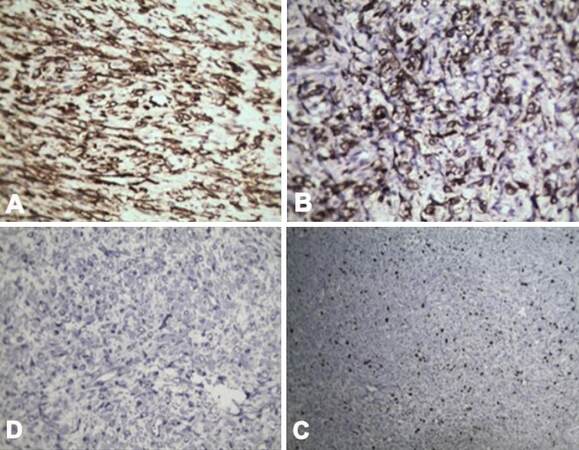
Our patient was a 1-year-old female child from Pakistan. She had decompensated chronic liver disease secondary to EHBA. She underwent primary LDLT at the age of 6 months at our center. Her mother was the donor. She had uneventful post transplantation recovery. At 3 months post liver transplantation (LT), routine USG revealed solid masses in the pelvis and in the sub hepatic region. Trucut biopsy of both the lesions revealed mesenchymal neoplasm. There was no evidence of malignancy. There was gradual increase in size of tumor on follow-up scans over the next 3 months [Figure 1A and B]. She reported back to our center for further evaluation of the intra-abdominal tumors. Review of previous biopsy slides and fresh trucut biopsies done at our center were inconclusive. A tumor board decision was made for surgical excision to get further information about the tumor and also relieve the mass effect in the abdomen. Elective laparotomy was planned. In laparotomy, a 15-cm lesion was seen in right sub diaphragmatic region pushing the hepatic flexure and graft liver which was also adherent to diaphragm [Figure 1C and D]. Another 7-cm firm mass was found adherent to omentum, proximal jejunum and jejunal mesentery. The tumor was removed along with a segment of jejunum. Minimal loculated ascites was also seen near the mass. Two other small tumors of approximately 2 cm each were seen attached to serosa of small bowel. All the macroscopically visible tumors were completely removed. HPE revealed IMT of the peritoneum with very mild malignant potential. Immunohistochemistry (IHC) was positive for anaplastic lymphoma kinase (ALK) antibodies [Figures 2 and 3]. Postoperative period was uneventful. She was discharged in a hemodynamically stable condition on postoperative day 10. After tumor board discussion, ceritinib was given as adjuvant therapy in view of ALK positivity. At 18 months follow-up, patient is recurrence free with good graft function.
Figure 1. Clinical pictures of the case report. (A) Cross sectional image showing the tumor along the cut surface of the graft liver; (B) coronal section of the PET-CT image showing PET avid lesions; (C) intra-operative picture showing one tumor near the cut surface of graft liver; (D) intra-operative picture showing the tumor masses along with the transplanted liver. PET: positron emission tomography; CT: computed tomography
Figure 2. Various magnifications (A: ×4; B: ×10; C: ×20; D: ×40) of haematoxylin and eosin staining showing liver parenchyma along with few malignant spindle cells
Discussion
The risk of de novo malignancy following LT is significantly higher than that of the general population. Skin, hematological, and colon cancers are common de novo malignancies after LT. Immunosuppression plays a major role in oncogenesis in the transplant population.[1] Other risk factors are hepatitis C virus infection, smoking, alcoholic cirrhosis, and sun exposure.[2]
IMTs are one of the rare groups of post-transplant malignancies. More than 200 cases have been described so far. Majority of them are after HSCT.[3,4] IMTs occur earlier in the post-transplant period ranging between 3 months and 2.5 years. Presenting complaints of patients are usually fever, weight loss and pain, along with mass related symptoms.[5] Sites of involvement are typically the abdomen, retroperitoneum and pelvis, followed by head, neck, upper respiratory tract, trunk and extremities. Increased morbidity may derive from its site in a vital organ or from aggressive treatment given due to a misdiagnosis of malignancy. Definitive histologic evaluation of the mass must be done to avoid unnecessary treatment-related complications. IMTs may rarely undergo malignant transformation, occasionally with distant metastases. The pathologic feature of IMTs is the proliferation of spindle cells associated with a variably dense polymorphic infiltrate of mononuclear inflammatory cells (e.g. lymphocytes, plasma cells, histiocytes, and occasional eosinophils). Coffin et al.[6] emphasized their neoplastic nature and proposed the use of the term inflammatory myofibroblastic tumor rather than inflammatory pseudo tumor. Other differential diagnoses include soft tissue sarcomas, gastrointestinal stromal tumors, lymphomas and other miscellaneous tumors. There is only one case report so far after deceased donor liver transplantation (DDLT).
In our case, multiple preoperative biopsies were not confirmatory and led to delay in treatment. Final excision biopsy was subjected to extensive evaluation with IHC panel which included c-KIT, ALK, CD117, CD 45, CK-7, neuron specific enolase, chromogranin, alpha fetal protein, desmin, actin. The tumor in this case was negative for other IHC markers including those specific for hepatocellular carcinoma, cholangiocarcinoma, neuroendocrine tumors, gastrointestinal stromal tumors and lymphomas. However, the tumor had classic characteristics of IMT along with positive IHC for anti-lymphoma kinase. Complete tumor excision is curative in most cases.[7] However recurrence at new sites can be prevalent. Ceritinibis a tyrosine kinase inhibitor accepted for treating tumors positive for ALK.[8] The patient was given ceritinib for 1 year. Follow-up with PET CT at 1 year revealed no recurrence. Hence ceritinib was discontinued. At 2 years, the child is disease free with good graft function.
In conclusion: (1) although IMTs are rare, it is a serious complication after liver transplantation; (2) only one case has been reported in the literature so far after liver transplantation that too after DDLT; (3) in this case report we have described the first case of IMT after LDLT; (4) definitive histologic evidence is essential in their diagnosis and differentiation from other malignant tumors; and (5) IHC based targeted therapy may be helpful as adjuvant.
Authors’ contributions
Doing surgery and writing of this manuscript: N. Selvakumar
Preparing the slides for histopathology: P. Saboti
Reporting of HPE: S. Kaul
Chief surgeon in charge of the case: S. Gupta
Financial support and sponsorship
None.
Conflicts of interest
There are no conflicts of interest.
Patient consent
Patient consent was received.
Ethics approval
Since it is not a study and just a report, ethical approval is not needed as per our hospital policies.
REFERENCES
1. Pillai AA. Management of de novo malignancies after liver transplantation. Transplant Rev (Orlando) 2015;29:38-41.
2. Piselli P, Busnach G, Fratino L, Citterio F, Ettorre GM, De Paoli P, Serraino D; Immunosuppression and Cancer Study Group. De novo malignancies after organ transplantation: focus on viral infections. Curr Mol Med 2013;13:1217-27.
3. Lykavieris P, Fabre M, Waguet J, Bernard O. Inflammatory pseudotumor after liver transplantation. J Pediatr Gastroenterol Nutr 2000;31:309-12.
4. Bronzino P, Abbo L, Bagnasco F, Barisone P, Dezzani C, Genovese AM, Iannucci P, Ippoliti M, Sacchi M, Aimo I. Intra-abdominal inflammatory myofibroblastic pseudotumor: case report and review of the literature. G Chir 2005;26:362-4.
5. Tufan A, Bahat G. Inflammatory pseudotumors after stem cell transplantation. Hematol Rep 2015;7:5848.
6. Lykavieris P, Fabre M, Waguet J, Bernard O. Inflammatory pseudotumor after liver transplantation. J Pediatr Gastroenterol Nutr 2000;31:309-12.
7. Vroobel K, Judson I, Dainton M, McCormick A, Fisher C, Thway K. ALK-positive inflammatory myofibroblastic tumor harboring ALK gene rearrangement, occurring after allogeneic stem cell transplant in an adult male. Pathol Res Pract 2016;212:743-6.
8. Chaudhary P. Mesenteric inflammatory myofibroblastic tumors. Ann Gastroenterol 2015;28:49-54.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Selvakumar N, Saboti P, Kaul S, Gupta S. First case report of inflammatory myofibroblastic tumor of peritoneal cavity in a living donor liver transplantation recipient. Hepatoma Res 2017;3:86-9. http://dx.doi.org/10.20517/2394-5079.2017.01
AMA Style
Selvakumar N, Saboti P, Kaul S, Gupta S. First case report of inflammatory myofibroblastic tumor of peritoneal cavity in a living donor liver transplantation recipient. Hepatoma Research. 2017; 3: 86-9. http://dx.doi.org/10.20517/2394-5079.2017.01
Chicago/Turabian Style
Selvakumar, Naganathan, Parul Saboti, Sumaid Kaul, Subash Gupta. 2017. "First case report of inflammatory myofibroblastic tumor of peritoneal cavity in a living donor liver transplantation recipient" Hepatoma Research. 3: 86-9. http://dx.doi.org/10.20517/2394-5079.2017.01
ACS Style
Selvakumar, N.; Saboti P.; Kaul S.; Gupta S. First case report of inflammatory myofibroblastic tumor of peritoneal cavity in a living donor liver transplantation recipient. Hepatoma. Res. 2017, 3, 86-9. http://dx.doi.org/10.20517/2394-5079.2017.01
About This Article
Copyright
Data & Comments
Data
 Cite This Article 1 clicks
Cite This Article 1 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.