Notch signaling in hepatocellular carcinoma: molecular targeting in an advanced disease
Abstract
The use of alternative therapeutic approaches in advanced carcinogenesis is a growing investigative base. One such cancer, primary liver cancer, is one of the most commonly occurring cancers worldwide and often presents in late stage disease consequently preventing traditional curative modalities. As a result, hepatocellular carcinoma (HCC), representing the majority of primary liver cancer, is the third most common cause of cancer-related deaths globally. Survival rates are linked to stage of presentation as well as concomitant cirrhosis limiting the 5-year survival in these patients to < 20%. Alternative strategies are in dire need as patients in this cohort have limited palliative options. Currently, sorafenib is the only approved systemic therapy; however, it has a limited survival advantage and low efficacy prompting the empirical need for further evaluation. Understanding of cancer therapy has led to an enhanced focus on the Notch pathway as a potential target for advanced HCC. Notch signaling is a critical component of development and cell fate and has been linked to various modalities including liver regeneration and as a key driver in carcinogenesis. In this review, we will provide a review of the current status of the Notch signaling in liver cancer and of Notch as an alternative potential strategy for advanced HCC.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is a significant health concern representing the sixth most common cancer globally.[1] Over the past 20 years, HCC has become one of the most frequent occurring tumors worldwide with the incidence in the United States steadily increasing.[2-5] In addition, coupled with an increase in the incidence, HCC mortality has also increased substantially. Currently, it is the third most common cause of cancer-related deaths throughout the world.
Approximately one-third of patients are amendable to curative therapy through the use of localized radiofrequency ablation or resection.[6,7] Moderate stage disease indicative of multifocal intrahepatic carcinogenesis has led to alternative approaches such as trans-arterial chemoembolization (TACE). TACE has provided a relatively efficacious avenue for patients in this category.[7-10] Patients progressing to or presenting as late stage disease have limited treatment options. Approximately, 70% of patients will initially or eventually present at this late stage. Consequently, this leads to a 5-year survival in patients with HCC of < 20%.[11] In addition, HCC is characteristically coupled with concomitant cirrhosis, further exacerbating disease morbidity and mortality.[12] Therefore, there is an urgent and critical need to expand alternative and effective approaches to these patients in advanced, nonresectable disease. This need for additional therapy and the evolving understanding of molecular pathways has led to a concentrated focus on therapeutic molecular targeting in many organ-specific cancers as well as HCC.
Several signaling pathways are of interest due to their specific oncogenic and/or tumor suppressor characteristics. For instance, the ras/raf pathway provides the only current approved therapeutic approach in advanced HCC through the use of sorafenib.[13,14] Other traditional pathways such as PI3k/Akt/mammalian target of rapamycin,[15-17] Wnt/β-catenin,[18] as well as Notch signaling[19-21] have been investigated. Further delineation into the manipulation of these pathways is critical for future alternative strategies for HCC. One of these pathways of interest is Notch signaling. As a functionally conserved pathway, it is involved in the regulation of several cellular properties including differentiation, proliferation, homeostasis and survival. First studied in Drosophila, Notch was linked to neural development. Future studies were able to identify the homology between species as well as accurately describe the Notch transmembrane receptor and provide evidence in its role as cellular regulator of differentiation, proliferation, and survival.[22-28] Continued work in the field of Notch signaling would inevitably showcase its role in a myriad of cellular processes centered on the development.
Notch signaling
The Notch signaling pathway consists of Notch receptors, ligands, negative and positive modifiers, and transcription factors. In mammals, these efficient modules have several members and the interplay between these molecules is not yet fully understood, but its role in several processes is being teased out including regulation of metabolism, inflammation, liver regeneration and repair.[29] Notch signaling is important from other conserved signaling pathways because its role in the mechanism of signal transduction is crucial. Compare to other intercellular signaling pathways such as Wnt, Hedgehog, and transforming growth factor-β, Notch is distinctive in several traits. First, the signaling of Notch is unique. It is comprised of both canonical and noncanonical signaling. The traditional canonical pathway occurs in a juxtacrine process that is unique to Notch. Cell-to-cell interaction is required for subsequent signaling. A transmitter cell releases one of the five major Notch ligands (Jagged 1, 2, and Delta-like 1, 3, 4) and binds to one of the four transmembrane Notch receptors (Notch 1-4) on the associate cell. Signaling is through several cleavage steps. The Notch receptor is cleaved by furin-like convertases in the trans-Golgi network, which results in two subunits of the mature/functional receptor. The extracellular Notch receptor subunit consists of a ligand-binding domain that is composed of epidermal growth factor-like repeats.[30] In addition, mammalian Notch-1, -2 and -3 receptors contain cytokine response regions and transcriptional activation domains.[30] Successful binding of the ligand to the representative receptor triggers a cleavage cascade of the Notch intracellular domain (NICD) via γ-secretase with NICD translocated to the nucleus.[31] NICD then binds with the RBP-Jκ family activating the complex as well as recruiting co-activator MAML1 that initiates transcription of Notch downstream targets including hairy enhancer of split, hairy enhancer of split with YRPW motif families, p21, and Sox-9.[32-36] Deactivation of the Notch signal is rapidly induced by phosphorylation and degradation. NICD is phosphorylated within the PEST domain by the CDK8 kinase and targeted for proteasomal degradation by E3 ubiquitin ligases that include Sel10/Fbw7.[37,38] Transcription activation of the ternary complex is disassembled and reset for the next round of signaling. With no second messenger to amplify its signal, deactivation is acute and tightly regulated.
Noncanonical Notch signaling involves a multiple of parallel pathways as cross-talk between these pathways dominates the influence of Notch through paracrine regulation.[31,39] One particular, well documented, example is the signaling of Notch and the Wnt/β-catenin pathways. Both pathways can act in synergistic concert through traditional Notch signaling or opposing interactions.[40] Conversely, antagonistic signaling is through noncanonical effects. A second crucial aspect particular to Notch signaling is the counteracting effects. Notch signals involve either the promotion or suppression of cell proliferation, cell death, and activation of differentiation programs. This happens in cells throughout development of the organism and during the maintenance of self-renewing adult tissues. Therefore, gain or loss of Notch signaling mechanisms has been directly linked to multiple human disorders. Even more conflicting in nature, opposing actions of Notch has been linked to similar disease processes in the liver.[41,42]
Notch signaling in the liver
The role of Notch signaling in the liver remained relatively unknown until the discovery and investigation of Alagille syndrome (AGS). As an autosomal dominant disease, AGS is characterized by ductopenia and cholestasis. Diminished development of intrahepatic bile ducts is the hallmark of AGS. Through genetic testing, near the turn of the century, it was demonstrated that mutations in the Jagged 1 gene and to a lesser extent Notch-2 led to AGS.[43-46] Therefore, it evidenced the role of Notch signaling in hepatogenesis, more specifically hepatic duct morphogenesis. Further research into the role of Notch in liver development focused on liver regeneration following injury. During injury and subsequent liver regeneration (i.e., partial liver resection) hepatocyte and cholangiocyte proliferative properties are often inhibited; therefore, precursor hepatic progenitor cells (HPCs) are activated in response to massive liver injury.[47-49] HPCs can differentiate or give rise to hepatocytes as well as cholangiocytes through Notch activation thus further strengthening the role of Notch in hepatogenesis and morphogenesis.[50-52] During repair and regeneration and at the height of concentrated HPC involvement, several pathways are activated to assist in morphogenesis including the Notch pathway. In addition, activation of the Notch pathway (specifically Notch-1 and -2 isoforms) is in concert with parallel pathways during regeneration.[40,53,54] These critical and emerging studies linking Notch activation to intrahepatic morphogenesis was a fundamental building block to the transition to reviewing the role of Notch in carcinogenesis.
Notch in carcinogenesis
As the expanding role of Notch signaling in the development of organogenesis continued, the role of Notch in carcinogenesis was ongoing. Notch-1 identification as an oncogene was first discovered through investigation into T-cell acute lymphoblastic leukemia (T-ALL). T-ALL gain of function mutations in Notch-1 led to overexpression and constitutive activation of Notch-1 receptor and thus enhanced proliferation.[55] In addition, the oncogenic ability of Notch was also exhibited in colorectal cancer.[56] According to Ambros, most Notch-mediated processes require a transient pulse of activity that in some cases lasts only as long as a fraction of the cell cycle degradation of the NICD.[57] This is of particular interest as Notch transduction has a 1:1 ratio of input to output without the presence of second messengers. Therefore, constitutive activation will provide constant transduction and thus aberrant proliferation. In addition to mutations in Notch-1 signaling leading to oncogenic enhancement, alterations in Notch-1 signaling also has led to changes in angiogenesis.[58]
The enhanced discoveries of the Notch signaling pathway lead to further advancement in additional solid tumors. Robinson et al.[59] studied the overexpression of Notch-1 and -2 fusion proteins in benign breast epithelial cells. Subsequent constitutive expression resulted in altered growth characteristics while the inhibition of Notch signaling reduced the growth of the Notch gene fusion-expressing breast cancer xenografts. Zender et al.[60] showed that overexpression of the Notch signaling pathway modulates the dysregulation of the oncogene cyclin E, resulting in the development of cholangiocellular carcinoma. In addition, inhibition of Notch activity blocks tumor cell proliferation and induces apoptosis in cholangiocellular carcinoma.[60] A retrospective analysis in oral squamous cell carcinoma (OSCC) showed that the Notch pathway was defective in 66% of patients and the studies of mechanism showed that the functional Notch-1 signaling inhibited proliferation of OSCC cell lines.[61]
Notch-1 dysregulation is not the only isoform involved in carcinogenesis. Studies have demonstrated that up regulation of the Notch-3 isoform was required for induction of p21 expression in senescent cells.[62] Inactivation of Notch-3 by γ-secretase inhibitor (GSI) or short interference RNA (siRNA) decreased cell proliferation and induced apoptosis in the chemoresistant ovarian cancer cells.[63] Finally, Lu et al.[64] have showed that the Notch-3 was positively correlated with Jagged-1 at the mRNA and protein levels. Therefore, they concluded that Notch-3 and Jagged-1 may play an important role in the initiation and proliferation of human nonfunctioning adenomas.[64]
Despite these early indications of Notch as a potential target for cancer therapy, the reality of Notch signaling in carcinogenesis remains opaque. Although there is growing evidence as to Notch acting as an oncogenic process, other cancers exhibit Notch’s role as a tumor suppressor in nature. For instance, in neuroendocrine tumors, Notch-1 acts as a tumor suppressor with overexpression leading to a reduction in cellular proliferation and growth.[26,65-67] Furthermore, despite thorough studies involving either the activation or inhibition of the Notch pathway in the modulation of carcinogenesis, there is limited data surrounding the expression of Notch receptors and their link with cancer. Additionally, noncanonical pathway activation of Notch further confuses and complicates the underlying roles of this pathway during times of aberrant cellular growth.
Targeting Notch in hepatocellular carcinoma
Despite promising results of Notch mediation in multiple organ-specific cancers, there is limited and conflicting data on Notch signaling in HCC. In several studies, Notch-1 acts as a tumor suppressor.[41,68] On the other hand, there is evidence that Notch is oncogenic in nature.[69-71] As time passes, growing evidence may indicate that although individual HCC signatures may include Notch as a tumor suppressor, the majority of HCC Notch mediation is through overexpression and oncogenic activation. For instance, Villanueva et al.[72] revealed that the conditional expression of NICD1 in a mouse model led to HCC in all test subjects within the 1st year. Biopsied tumors represented varying stages in the mice test group. Moreover, overexpression of Notch-1 was closely linked to insulin-like growth factor 2 and Sox-9 expression levels and interestingly, the NICD1 conditionally active mice genetic signature was evidenced in a subset of human patients with HCC.[72] Further studies have shifted the tide toward Notch acting as an oncogene in liver carcinogenesis. Sox-9, a downstream target of Notch signaling cascade, is linked to cellular proliferation and carries a worse prognosis. Overexpression of Sox-9 leads to a transition to HPC-type activity and consequently less differentiated cell types.[73]
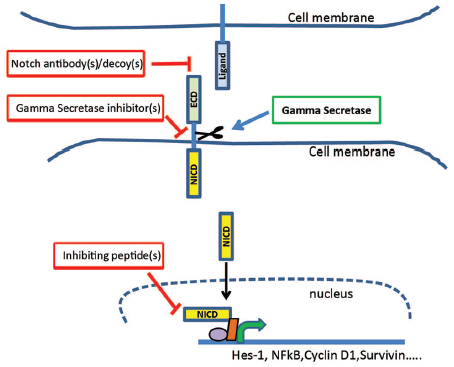
As we advance our understanding of the Notch signaling pathway, initial studies including several early phase I clinical trials are underway in various stepwise components within the pathway. Given the intricacy of the canonical Notch pathway, it comes as no surprise that there are multiple avenues to target Notch signaling [Figure 1]. Targeting either the ligands and/or the receptors, inhibiting cleavage of the active NICD, and preventing transcription of downstream targets are the major targeted aims in Notch mediation. In this review, we will briefly discuss the most common techniques aimed at inhibiting Notch signaling.
Figure 1. Canonical Notch signaling pathway and potential sites of inhibition. The Notch pathway is primed through cell-to-cell interaction distinguishing it from other regulatory pathways. Following ligand secretion from a transmitting cell, the ligand binds to one of the four Notch receptors on the receiver cell. Ligand-receptor binding facilitates the cleavage of the intracellular component of the transmembrane receptor via γ-secretase. Successful cleavage activates the Notch intracellular component domain (NICD) which translocates to the nucleus where is regulated a host of transcription factors. Given the intricacy of the pathway, there are multiple key regulatory steps poised for targeted therapy. First, monoclonal antibody and decoy administration at both the Notch ligand and receptor is in preliminary, preclinical investigation. Second, γ-secretase inhibitors are the most studied target within the pathway; however, there is limited data within hepatocellular carcinoma. Finally, inactivating transcriptional peptides are a novel trend focused on inhibiting the canonical transcription mediated by NICD
The most studied area is inhibition of GSI and the subsequent release of NICD. In fact, GSI examination is not inclusive to HCC. Rather, the extensive research of Notch signaling in neural development has led to GSI application for Alzheimer’s disease.[74] There has been countless preclinical and phase I clinical trials examining the efficacy and effectiveness of GSI mediation in many cancer types.[75-77] Unfortunately, to date, there is no phase I evidence of the role of GSIs in HCC. Moreover, given the pan-inhibition nature of GSIs, the toxicity profiles are relatively disconcerting given the off target effects, especially intestinal adverse effects specifically through down regulation of Notch-1 and -2 isoforms.[29]
As a result of the nonspecific inhibition of GSIs, alternative strategies should be considered. One particular area of increasing interest is the use of monoclonal antibodies and decoys at both the ligand and receptor sites. Antibody and decoy (competitive antagonist) application has a more specific efficacy, thus limiting the dose-escalated toxicities and potentially providing a concentrated result. There are multiple monoclonal antibodies currently being tested in preclinical studies. Notch-1 receptor antibodies have shown promising results.[78,79] Both Notch-2 and Notch-3 antibodies have transitioned to phase I clinical trials.[79,80] In addition to Notch receptor blockade, antibodies against Notch signaling ligands have been investigated. Delta-like ligand four antibodies have shown interesting results from multiple avenues.[58,81,82] Similarly, decoys provide excellent Notch inhibition and act as a competitive antagonist either at the Notch receptor or the ligand binding sites. Notch-1 decoys have been studied as well as Jagged-1 ligand decoys.[83,84] Finally, prevention of NICD-mediated transcription is a novel process to modulate carcinogenesis. Peptides that block the transcription of NICD provide interesting applications to Notch signal inhibition.[75] In addition to this review of potential Notch mediation through the alteration of multiple events, Espinoza and Miele[75] recently compiled a comprehensive table and analysis including a majority of current preclinical and clinical studies using a myriad of Notch inhibitors that further details the current effectiveness of Notch alteration.
Traditional mechanisms of Notch inhibition have and will continue to be thoroughly investigated; however, there is growing interest in targeted gene inhibition, more specifically in the context of Notch signaling. Historically, AGS and the mechanism of hepatogenesis were further delineated through Notch-2 gene manipulation; therefore, an approach to targeting specific genes within the Notch pathway may provide additional support in lieu of traditional Notch inhibitors. There are several approaches to gene silencing, two of which are frequently used and include using a small hairpin RNA or short hairpin RNA (shRNA) or siRNA. Mao et al.[85] reported that the shRNA mediated knock-down of Notch-1 inhibited the breast cancer cell line MCF-7’s proliferation and induced cell apoptosis through multiple mechanistic actions. One in particular, the down regulation of the anti-apoptotic protein nuclear factor-kappa B, proved effective and enhanced the anti-tumorigenic effect when combined with traditional chemotherapeutic agents such as paclitaxel. Finally, Mao et al.[85] were able to translate this to an in vivo study evidencing that genetic knockout of Notch-1 abrogated tumor xenograft growth. Zhao et al.[86] reported that the knockdown of Notch-1 by RNA interference suppressed Akt activation, reduced glioma cell growth rate and induce cell apoptosis.
Notch-1 deletion has also been studied in HCC. Sun and colleagues investigated that knockout of Notch-1 inhibited cell proliferation and significantly suppressed tumor formation of L02/HBx cells in a BALB/c nude mouse model in vivo through activation of apoptotic caspase cascades. In addition, they observed that this blockade arrest the cell cycle in the G0/G1 phase through the down regulation of cyclin D1, CDK4, E2F1 and the up regulation of p21.[87] Wang et al.[88] suggested that the inhibition of Notch1 by shRNA significantly suppressed the growth of HBx transformed human hepatic cells through G0/G1 cell cycle arrest and apoptosis. The mechanism, they suggested, may be linked to the promoted expression of P16 and decreased expression of Bcl-2.[88]
Finally, the investigation into microRNAs as a potential strategy is growing in interest. MicroRNAs are small regulators of both post-translational and post-transcriptional markers. They are often at the center of abrogation in many cancer types.[89,90] Given their stability, they are potential candidates for use in combination studies. For instance, there is increasing data on the use of microRNAs sensitizing HCC to traditional chemotherapy.[91,92] In addition, genetic profiling of microRNAs in patients with HCC will assist as an alternative and supportive strategy in terms of disease-free progression and overall survival. For example, microRNA-224 expression is associated with a better prognosis and further evaluation into this subtype is currently ongoing.[93,94]
Despite the plethora of early investigations into Notch inhibition, there are concerns that need to be addressed moving forward. First, there are relatively limited studies advancing in HCC research. As of this publication, there are no clinical trials utilizing Notch inhibition as an alternative strategy for HCC. In addition, in other solid tumor studies, no trial has advanced past phase I. Perhaps we are in the early stages of development, but given the stagnant advancement additional approaches should be addressed.
Transition to the future
The exciting and staggering concept of Notch signaling is that it is still in the infantile stages of development. The majority of evolution in the understanding of this pathway has come within the last 20-30 years. Within that time, there have been novel and potentially ground-breaking investigations into the role of Notch not only in HCC, but other cancer types. In addition, the study of Notch mediation has radiated toward different fields of medicine with the intent of delineating the roles of isoform-specific NICD. Additionally, the role of cellular homeostasis has interpretive results in a myriad of clinical and basic science indications and perhaps Notch will be at the forefront of these studies. However, the role of Notch in carcinogenesis, albeit, counterintuitive, is both exciting and complex. The early results prove modulation of this pathway could aid in the care of advanced, resistant, and aggressive cancer types. These Notch-based strategies will continue to be evaluated and will also be combined with other pathway mitigation to reduce toxicity profiles, as well as the chemoresistance. Combination with approved and current strategies will further the understanding and commitment to providing alternative and efficacious treatment options to patients with HCC.
Financial support and sponsorship
This review was supported by the Medical College of Wisconsin Dean’s Development Program, the Medical College of Wisconsin Research Affairs Committee, the Medical College of Wisconsin Digestive Disease Center Fund, and the Froedtert Hospital Foundation.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. European Association For The Study Of The Liver, European Organisation For Research And Treatment Of Cancer. EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 2012;56:908-43.
2. Altekruse SF, Henley SJ, Cucinelli JE, McGlynn KA. Changing hepatocellular carcinoma incidence and liver cancer mortality rates in the United States. Am J Gastroenterol 2014;109:542-53.
4. Schütte K, Bornschein J, Malfertheiner P. Hepatocellular carcinoma – epidemiological trends and risk factors. Dig Dis 2009;27:80-92.
6. McGlynn KA, Tsao L, Hsing AW, Devesa SS, Fraumeni JF Jr. International trends and patterns of primary liver cancer. Int J Cancer 2001;94:290-6.
7. Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: chemoembolization improves survival. Hepatology 2003;37:429-42.
8. Bruix J, Sherman M. Practice Guidelines Committee, American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma. Hepatology 2005;42:1208-36.
9. Sergio A, Cristofori C, Cardin R, Pivetta G, Ragazzi R, Baldan A, Girardi L, Cillo U, Burra P, Giacomin A, Farinati F. Transcatheter arterial chemoembolization (TACE) in hepatocellular carcinoma (HCC): the role of angiogenesis and invasiveness. Am J Gastroenterol 2008;103:914-21.
10. Miura JT, Gamblin TC. Transarterial chemoembolization for primary liver malignancies and colorectal liver metastasis. Surg Oncol Clin N Am 2015;24:149-66.
11. Pons F, Varela M, Llovet JM. Staging systems in hepatocellular carcinoma. HPB (Oxford) 2005;7:35-41.
12. McGlynn KA, London WT. The global epidemiology of hepatocellular carcinoma: present and future. Clin Liver Dis 2011;15:223-43, vii.
13. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Haussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J; SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359:378-90.
14. Villanueva A, Llovet JM. Targeted therapies for hepatocellular carcinoma. Gastroenterology 2011;140:1410-26.
15. Nakanishi K, Sakamoto M, Yamasaki S, Todo S, Hirohashi S. Akt phosphorylation is a risk factor for early disease recurrence and poor prognosis in hepatocellular carcinoma. Cancer 2005;103:307-12.
16. Vanhaesebroeck B, Stephens L, Hawkins P. PI3K signalling: the path to discovery and understanding. Nat Rev Mol Cell Biol 2012;13:195-203.
17. Vivanco I, Sawyers CL. The phosphatidylinositol 3-Kinase AKT pathway in human cancer. Nat Rev Cancer 2002;2:489-501.
18. Lachenmayer A, Alsinet C, Savic R, Cabellos L, Toffanin S, Hoshida Y, Villanueva A, Minguez B, Newell P, Tsai HW, Barretina J, Thung S, Ward SC, Bruix J, Mazzaferro V, Schwartz M, Friedman SL, Llovet JM. Wnt-pathway activation in two molecular classes of hepatocellular carcinoma and experimental modulation by sorafenib. Clin Cancer Res 2012;18:4997-5007.
19. Ranganathan P, Weaver KL, Capobianco AJ. Notch signalling in solid tumours: a little bit of everything but not all the time. Nat Rev Cancer 2011;11:338-51.
20. Razumilava N, Gores GJ. Notch-driven carcinogenesis: the merging of hepatocellular cancer and cholangiocarcinoma into a common molecular liver cancer subtype. J Hepatol 2013;58:1244-5.
21. Rose SL, Kunnimalaiyaan M, Drenzek J, Seiler N. Notch 1 signaling is active in ovarian cancer. Gynecol Oncol 2010;117:130-3.
22. Austin J, Kimble J. glp-1 is required in the germ line for regulation of the decision between mitosis and meiosis in C. elegans. Cell 1987;51:589-99.
23. Cabrera CV. The generation of cell diversity during early neurogenesis in Drosophila. Development 1992;115:893-901.
24. Coffman C, Harris W, Kintner C. Xotch, the Xenopus homolog of Drosophila notch. Science 1990;249:1438-41.
25. Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD, Sklar J. TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell 1991;66:649-61.
26. Kunnimalaiyaan M, Chen H. Tumor suppressor role of Notch-1 signaling in neuroendocrine tumors. Oncologist 2007;12:535-42.
27. Priess JR, Schnabel H, Schnabel R. The glp-1 locus and cellular interactions in early C. elegans embryos. Cell 1987;51:601-11.
28. Yamamoto S, Schulze KL, Bellen HJ. Introduction to Notch signaling. Methods Mol Biol 2014;1187:1-14.
29. Morell CM, Strazzabosco M. Notch signaling and new therapeutic options in liver disease. J Hepatol 2014;60:885-90.
31. Andersson ER, Lendahl U. Therapeutic modulation of Notch signalling – are we there yet? Nat Rev Drug Discov 2014;13:357-78.
32. Allenspach EJ, Maillard I, Aster JC, Pear WS. Notch signaling in cancer. Cancer Biol Ther 2002;1:466-76.
33. Fortini ME. Notch signaling: the core pathway and its posttranslational regulation. Dev Cell 2009;16:633-47.
34. Geisler F, Strazzabosco M. Emerging roles of Notch signaling in liver disease. Hepatology 2015;61:382-92.
36. Strazzabosco M, Fabris L. Notch signaling in hepatocellular carcinoma: guilty in association. Gastroenterology 2012;143:1430-4.
37. O'Neil J, Grim J, Strack P, Rao S, Tibbitts D, Winter C, Hardwick J, Welcker M, Meijerink JP, Pieters R, Draetta G, Sears R, Clurman BE, Look AT. FBW7 mutations in leukemic cells mediate NOTCH pathway activation and resistance to gamma-secretase inhibitors. J Exp Med 2007;204:1813-24.
38. Thompson BJ, Buonamici S, Sulis ML, Palomero T, Vilimas T, Basso G, Ferrando A, Aifantis I. The SCFFBW7 ubiquitin ligase complex as a tumor suppressor in T cell leukemia. J Exp Med 2007;204:1825-35.
39. D'Souza B, Meloty-Kapella L, Weinmaster G. Canonical and non-canonical Notch ligands. Curr Top Dev Biol 2010;92:73-129.
40. Boulter L, Govaere O, Bird TG, Radulescu S, Ramachandran P, Pellicoro A, Ridgway RA, Seo SS, Spee B, Van Rooijen N, Sansom OJ, Iredale JP, Lowell S, Roskams T, Forbes SJ. Macrophage-derived Wnt opposes Notch signaling to specify hepatic progenitor cell fate in chronic liver disease. Nat Med 2012;18:572-9.
41. Viatour P, Ehmer U, Saddic LA, Dorrell C, Andersen JB, Lin C, Zmoos AF, Mazur PK, Schaffer BE, Ostermeier A, Vogel H, Sylvester KG, Thorgeirsson SS, Grompe M, Sage J. Notch signaling inhibits hepatocellular carcinoma following inactivation of the RB pathway. J Exp Med 2011;208:1963-76.
42. Wu G, Wilson G, George J, Qiao L. Modulation of Notch signaling as a therapeutic approach for liver cancer. Curr Gene Ther 2015;15:171-81.
43. Oda T, Elkahloun AG, Pike BL, Okajima K, Krantz ID, Genin A, Piccoli DA, Meltzer PS, Spinner NB, Collins FS, Chandrasekharappa SC. Mutations in the human Jagged1 gene are responsible for Alagille syndrome. Nat Genet 1997;16:235-42.
44. Li PH, Shu SG, Yang CH, Lo FC, Wen MC, Chi CS. Alagille syndrome with interstitial 20p deletion derived from maternal ins (7;20). Am J Med Genet 1996;63:537-41.
45. Li L, Krantz ID, Deng Y, Genin A, Banta AB, Collins CC, Qi M, Trask BJ, Kuo WL, Cochran J, Costa T, Pierpont ME, Rand EB, Piccoli DA, Hood L, Spinner NB. Alagille syndrome is caused by mutations in human Jagged1, which encodes a ligand for Notch1. Nat Genet 1997;16:243-51.
46. McDaniell R, Warthen DM, Sanchez-Lara PA, Pai A, Krantz ID, Piccoli DA, Spinner NB. NOTCH2 mutations cause Alagille syndrome, a heterogeneous disorder of the notch signaling pathway. Am J Hum Genet 2006;79:169-73.
48. Riehle KJ, Dan YY, Campbell JS, Fausto N. New concepts in liver regeneration. J Gastroenterol Hepatol 2011;26 suppl 1:203-12.
49. Michalopoulos GK. Liver regeneration: alternative epithelial pathways. Int J Biochem Cell Biol 2011;43:173-9.
50. Fiorotto R, Raizner A, Morell CM, Torsello B, Scirpo R, Fabris L, Spirli C, Strazzabosco M. Notch signaling regulates tubular morphogenesis during repair from biliary damage in mice. J Hepatol 2013;59:124-30.
51. Morell CM, Fiorotto R, Fabris L, Strazzabosco M. Notch signalling beyond liver development: emerging concepts in liver repair and oncogenesis. Clin Res Hepatol Gastroenterol 2013;37:447-54.
52. Tanimizu N, Miyajima A. Notch signaling controls hepatoblast differentiation by altering the expression of liver-enriched transcription factors. J Cell Sci 2004;117:3165-74.
53. Chen Y, Choi SS, Michelotti GA, Chan IS, Swiderska-Syn M, Karaca GF, Xie G, Moylan CA, Garibaldi F, Premont R, Suliman HB, Piantadosi CA, Diehl AM. Hedgehog controls hepatic stellate cell fate by regulating metabolism. Gastroenterology 2012;143:1319-29.
54. Xie G, Karaca G, Swiderska-Syn M, Michelotti GA, Krüger L, Chen Y, Premont RT, Choi SS, Diehl AM. Cross-talk between Notch and Hedgehog regulates hepatic stellate cell fate in mice. Hepatology 2013;58:1801-13.
55. Weng AP, Ferrando AA, Lee W, Morris JP 4th, Silverman LB, Sanchez-Irizarry C, Blacklow SC, Look AT, Aster JC. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science 2004;306:269-71.
56. van Es JH, van Gijn ME, Riccio O, van den Born M, Vooijs M, Begthel H, Cozijnsen M, Robine S, Winton DJ, Radtke F, Clevers H. Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature 2005;435:959-63.
57. Ambros V. Cell cycle-dependent sequencing of cell fate decisions in Caenorhabditis elegans vulva precursor cells. Development 1999;126:1947-56.
58. Ridgway J, Zhang G, Wu Y, Stawicki S, Liang WC, Chanthery Y, Kowalski J, Watts RJ, Callahan C, Kasman I, Singh M, Chien M, Tan C, Hongo JA, de Sauvage F, Plowman G, Yan M. Inhibition of Dll4 signalling inhibits tumour growth by deregulating angiogenesis. Nature 2006;444:1083-7.
59. Robinson DR, Kalyana-Sundaram S, Wu YM, Shankar S, Cao X, Ateeq B, Asangani IA, Iyer M, Maher CA, Grasso CS, Lonigro RJ, Quist M, Siddiqui J, Mehra R, Jing X, Giordano TJ, Sabel MS, Kleer CG, Palanisamy N, Natrajan R, Lambros MB, Reis-Filho JS, Kumar-Sinha C, Chinnaiyan AM. Functionally recurrent rearrangements of the MAST kinase and Notch gene families in breast cancer. Nat Med 2011;17:1646-51.
60. Zender S, Nickeleit I, Wuestefeld T, Sörensen I, Dauch D, Bozko P, El-Khatib M, Geffers R, Bektas H, Manns MP, Gossler A, Wilkens L, Plentz R, Zender L, Malek NP. A critical role for notch signaling in the formation of cholangiocellular carcinomas. Cancer Cell 2013;23:784-95.
61. Pickering CR, Zhang J, Yoo SY, Bengtsson L, Moorthy S, Neskey DM, Zhao M, Ortega Alves MV, Chang K, Drummond J, Cortez E, Xie TX, Zhang D, Chung W, Issa JP, Zweidler-McKay PA, Wu X, El-Naggar AK, Weinstein JN, Wang J, Muzny DM, Gibbs RA, Wheeler DA, Myers JN, Frederick MJ. Integrative genomic characterization of oral squamous cell carcinoma identifies frequent somatic drivers. Cancer Discov 2013;3:770-81.
62. Cui H, Kong Y, Xu M, Zhang H. Notch3 functions as a tumor suppressor by controlling cellular senescence. Cancer Res 2013;73:3451-9.
63. Rahman MT, Nakayama K, Rahman M, Katagiri H, Katagiri A, Ishibashi T, Ishikawa M, Iida K, Nakayama S, Otsuki Y, Miyazaki K. Notch3 overexpression as potential therapeutic target in advanced stage chemoresistant ovarian cancer. Am J Clin Pathol 2012;138:535-44.
64. Lu R, Gao H, Wang H, Cao L, Bai J, Zhang Y. Overexpression of the Notch3 receptor and its ligand Jagged1 in human clinically non-functioning pituitary adenomas. Oncol Lett 2013;5:845-51.
65. Greenblatt DY, Vaccaro AM, Jaskula-Sztul R, Ning L, Haymart M, Kunnimalaiyaan M, Chen H. Valproic acid activates notch-1 signaling and regulates the neuroendocrine phenotype in carcinoid cancer cells. Oncologist 2007;12:942-51.
66. Nakakura EK, Sriuranpong VR, Kunnimalaiyaan M, Hsiao EC, Schuebel KE, Borges MW, Jin N, Collins BJ, Nelkin BD, Chen H, Ball DW. Regulation of neuroendocrine differentiation in gastrointestinal carcinoid tumor cells by notch signaling. J Clin Endocrinol Metab 2005;90:4350-6.
67. Ning L, Greenblatt DY, Kunnimalaiyaan M, Chen H. Suberoyl bis-hydroxamic acid activates Notch-1 signaling and induces apoptosis in medullary thyroid carcinoma cells. Oncologist 2008;13:98-104.
68. Croquelois A, Blindenbacher A, Terracciano L, Wang X, Langer I, Radtke F, Heim MH. Inducible inactivation of Notch1 causes nodular regenerative hyperplasia in mice. Hepatology 2005;41:487-96.
69. Giovannini C, Baglioni M, Baron Toaldo M, Ventrucci C, D'Adamo S, Cipone M, Chieco P, Gramantieri L, Bolondi L. Notch3 inhibition enhances sorafenib cytotoxic efficacy by promoting GSK3b phosphorylation and p21 down-regulation in hepatocellular carcinoma. Oncotarget 2013;4:1618-31.
70. Giovannini C, Gramantieri L, Chieco P, Minguzzi M, Lago F, Pianetti S, Ramazzotti E, Marcu KB, Bolondi L. Selective ablation of Notch3 in HCC enhances doxorubicin's death promoting effect by a p53 dependent mechanism. J Hepatol 2009;50:969-79.
71. Gramantieri L, Giovannini C, Lanzi A, Chieco P, Ravaioli M, Venturi A, Grazi GL, Bolondi L. Aberrant Notch3 and Notch4 expression in human hepatocellular carcinoma. Liver Int 2007;27:997-1007.
72. Villanueva A, Alsinet C, Yanger K, Hoshida Y, Zong Y, Toffanin S, Rodriguez-Carunchio L, Sole M, Thung S, Stanger BZ, Llovet JM. Notch signaling is activated in human hepatocellular carcinoma and induces tumor formation in mice. Gastroenterology 2012;143:1660-9.
73. Guo X, Xiong L, Sun T, Peng R, Zou L, Zhu H, Zhang J, Li H, Zhao J. Expression features of SOX9 associate with tumor progression and poor prognosis of hepatocellular carcinoma. Diagn Pathol 2012;7:44.
74. Siemers ER, Quinn JF, Kaye J, Farlow MR, Porsteinsson A, Tariot P, Zoulnouni P, Galvin JE, Holtzman DM, Knopman DS, Satterwhite J, Gonzales C, Dean RA, May PC. Effects of a gamma-secretase inhibitor in a randomized study of patients with Alzheimer disease. Neurology 2006;66:602-4.
76. Tammam J, Ware C, Efferson C, O'Neil J, Rao S, Qu X, Gorenstein J, Angagaw M, Kim H, Kenific C, Kunii K, Leach KJ, Nikov G, Zhao J, Dai X, Hardwick J, Scott M, Winter C, Bristow L, Elbi C, Reilly JF, Look T, Draetta G, Van der Ploeg L, Kohl NE, Strack PR, Majumder PK. Down-regulation of the Notch pathway mediated by a gamma-secretase inhibitor induces anti-tumour effects in mouse models of T-cell leukaemia. Br J Pharmacol 2009;158:1183-95.
77. Wei P, Walls M, Qiu M, Ding R, Denlinger RH, Wong A, Tsaparikos K, Jani JP, Hosea N, Sands M, Randolph S, Smeal T. Evaluation of selective gamma-secretase inhibitor PF-03084014 for its antitumor efficacy and gastrointestinal safety to guide optimal clinical trial design. Mol Cancer Ther 2010;9:1618-28.
78. Aste-Amézaga M, Zhang N, Lineberger JE, Arnold BA, Toner TJ, Gu M, Huang L, Vitelli S, Vo KT, Haytko P, Zhao JZ, Baleydier F, L'Heureux S, Wang H, Gordon WR, Thoryk E, Andrawes MB, Tiyanont K, Stegmaier K, Roti G, Ross KN, Franlin LL, Wang H, Wang F, Chastain M, Bett AJ, Audoly LP, Aster JC, Blacklow SC, Huber HE. Characterization of Notch1 antibodies that inhibit signaling of both normal and mutated Notch1 receptors. PLoS One 2010;5:e9094.
79. Wu Y, Cain-Hom C, Choy L, Hagenbeek TJ, de Leon GP, Chen Y, Finkle D, Venook R, Wu X, Ridgway J, Schahin-Reed D, Dow GJ, Shelton A, Stawicki S, Watts RJ, Zhang J, Choy R, Howard P, Kadyk L, Yan M, Zha J, Callahan CA, Hymowitz SG, Siebel CW. Therapeutic antibody targeting of individual Notch receptors. Nature 2010;464:1052-7.
80. Li K, Li Y, Wu W, Gordon WR, Chang DW, Lu M, Scoggin S, Fu T, Vien L, Histen G, Zheng J, Martin-Hollister R, Duensing T, Singh S, Blacklow SC, Yao Z, Aster JC, Zhou BB. Modulation of Notch signaling by antibodies specific for the extracellular negative regulatory region of NOTCH3. J Biol Chem 2008;283:8046-54.
81. Noguera-Troise I, Daly C, Papadopoulos NJ, Coetzee S, Boland P, Gale NW, Lin HC, Yancopoulos GD, Thurston G. Blockade of Dll4 inhibits tumour growth by promoting non-productive angiogenesis. Nature 2006;444:1032-7.
82. Scehnet JS, Jiang W, Kumar SR, Krasnoperov V, Trindade A, Benedito R, Djokovic D, Borges C, Ley EJ, Duarte A, Gill PS. Inhibition of Dll4-mediated signaling induces proliferation of immature vessels and results in poor tissue perfusion. Blood 2007;109:4753-60.
83. Funahashi Y, Hernandez SL, Das I, Ahn A, Huang J, Vorontchikhina M, Sharma A, Kanamaru E, Borisenko V, Desilva DM, Suzuki A, Wang X, Shawber CJ, Kandel JJ, Yamashiro DJ, Kitajewski J. A notch1 ectodomain construct inhibits endothelial notch signaling, tumor growth, and angiogenesis. Cancer Res 2008;68:4727-35.
84. Small D, Kovalenko D, Kacer D, Liaw L, Landriscina M, Di Serio C, Di Serio C, Prudovsky I, Maciag T. Soluble Jagged 1 represses the function of its transmembrane form to induce the formation of the Src-dependent chord-like phenotype. J Biol Chem 2001;276:32022-30.
85. Mao J, Song B, Shi Y, Wang B, Fan S, Yu X, Tang J, Li L. ShRNA targeting Notch1 sensitizes breast cancer stem cell to paclitaxel. Int J Biochem Cell Biol 2013;45:1064-73.
86. Zhao N, Guo Y, Zhang M, Lin L, Zheng Z. Akt-mTOR signaling is involved in Notch-1-mediated glioma cell survival and proliferation. Oncol Rep 2010;23:1443-7.
87. Sun Q, Wang R, Wang Y, Luo J, Wang P, Cheng B. Notch1 is a potential therapeutic target for the treatment of human hepatitis B virus X protein-associated hepatocellular carcinoma. Oncol Rep 2014;31:933-9.
88. Wang F, Xia X, Wang J, Sun Q, Luo J, Cheng B. Notch1 signaling contributes to the oncogenic effect of HBx on human hepatic cells. Biotechnol Lett 2013;35:29-37.
89. Gramantieri L, Fornari F, Callegari E, Sabbioni S, Lanza G, Croce CM, Bolondi L, Negrini M. MicroRNA involvement in hepatocellular carcinoma. J Cell Mol Med 2008;12:2189-204.
91. Xu Y, Xia F, Ma L, Shan J, Shen J, Yang Z, Liu J, Cui Y, Bian X, Bie P, Qian C. MicroRNA-122 sensitizes HCC cancer cells to adriamycin and vincristine through modulating expression of MDR and inducing cell cycle arrest. Cancer Lett 2011;310:160-9.
92. Bai S, Nasser MW, Wang B, Hsu SH, Datta J, Kutay H, Yadav A, Nuovo G, Kumar P, Ghoshal K. MicroRNA-122 inhibits tumorigenic properties of hepatocellular carcinoma cells and sensitizes these cells to sorafenib. J Biol Chem 2009;284:32015-27.
93. Wang Y, Lee AT, Ma JZ, Wang J, Ren J, Yang Y, Tantoso E, Li KB, Ooi LL, Tan P, Lee CG. Profiling microRNA expression in hepatocellular carcinoma reveals microRNA-224 up-regulation and apoptosis inhibitor-5 as a microRNA-224-specific target. J Biol Chem 2008;283:13205-15.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Sokolowski KM, Balamurugan M, Kunnimalaiyaan S, Gamblin TC, Kunnimalaiyaan M. Notch signaling in hepatocellular carcinoma: molecular targeting in an advanced disease. Hepatoma Res 2015;1:11-8. http://dx.doi.org/10.4103/2394-5079.154132
AMA Style
Sokolowski KM, Balamurugan M, Kunnimalaiyaan S, Gamblin TC, Kunnimalaiyaan M. Notch signaling in hepatocellular carcinoma: molecular targeting in an advanced disease. Hepatoma Research. 2015; 1: 11-8. http://dx.doi.org/10.4103/2394-5079.154132
Chicago/Turabian Style
Sokolowski, Kevin M., Mariappan Balamurugan, Selvi Kunnimalaiyaan, T. Clark Gamblin, Muthusamy Kunnimalaiyaan. 2015. "Notch signaling in hepatocellular carcinoma: molecular targeting in an advanced disease" Hepatoma Research. 1: 11-8. http://dx.doi.org/10.4103/2394-5079.154132
ACS Style
Sokolowski, KM.; Balamurugan M.; Kunnimalaiyaan S.; Gamblin TC.; Kunnimalaiyaan M. Notch signaling in hepatocellular carcinoma: molecular targeting in an advanced disease. Hepatoma. Res. 2015, 1, 11-8. http://dx.doi.org/10.4103/2394-5079.154132
About This Article
Copyright
Data & Comments
Data
 Cite This Article 8 clicks
Cite This Article 8 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.