Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells
Abstract
Aim: Aflatoxin B1 (AFB1) and fumonisin B1 (FB1) are important food-borne mycotoxins. Co-contamination of foodstuffs with these two mycotoxins is well-known and has been implicated in a possible development of hepatocellular carcinoma in humans living in regions of the world where exposures to these mycotoxins in grain are greatest. The aim of the current study was to evaluate the potential protective effects of an aqueous extract of Cochorus olitorius (C. olitorius, moroheiya) against cytotoxicity of AFB1 and/or FB1 in H4IIE-luc rat hepatoma cells, using assays to measure cell viability and disruption of DNA integrity. Although this transactivation assay was originally developed to specifically respond to aryl hydrocarbon agonists, this cell line was used because of its hepatic origin.
Methods: H4IIE-luc cells were incubated with different concentrations of AFB1 and/or FB1 for 24 and 48 h with or without aqueous extract of C. olitorius.
Results: Both mycotoxins decreased cell viability and increased DNA damage. Cytotoxicity was more pronounced when cells were exposed simultaneously to AFB1 and FB1.
Conclusion: Aqueous extract of C. olitorius protected cells against cytotoxicity of mycotoxins. C. olitorius contains a water-soluble, natural chemo-preventative agent for cancer that should be isolated and identified.
Keywords
Introduction
Co-occurrence of various mycotoxins in foodstuffs and animal feed is common because each toxigenic fungus can produce more than one mycotoxin and foodstuff can be colonized by several fungi either while growing in the field or during storage or transport.[1] Processed products are often composed of various raw materials which might be contaminated with mycotoxins.[2] Poor harvest practices and inadequate conditions during drying, handling, packaging, storing and transporting can contribute to the growth of fungi and an increased risk of production of mycotoxins.[3] The importance of co-occurrence of mycotoxins lies in the changes that might occur in the combined toxicity of mycotoxins.[4] In addition, the existence of relationships in the occurrences of mycotoxins allows predictions of the presence of individual mycotoxins from the presence of others.[1]
Among these mycotoxins, aflatoxin B1 (AFB1) is the predominant contaminant in cereals and oilseed and presents a significant risk,[5] due to being hepatotoxic and carcinogenic to humans and animals.[4,6-8] AFB1 is classified by the International Agency of Research on Cancer (IARC) as a Group 1 carcinogen.[9] This mycotoxin is also mutagenic, teratogenic, and immunosuppressive in farm, and laboratory animals,[10-12] and primarily affects cell-mediated immunity.[13] AFB1 is also able to induce reactive oxygen species (ROS),[8,14-16] possibly requiring activation of cytochrome P450.
Fumonisins, mainly produced by Fusarium verticillioides and F. proliferatum, are mycotoxins commonly found on corn. The most toxic and abundant of these is fumonisin B1 (FB1), which causes esophageal and hepatic cancer in humans and liver and kidney cancer in rodents.[17-19] IARC evaluated FB1, and classified it as probably carcinogenic to humans (Group 2B).[20] Moreover, FB1 modulates immunity in animals and decreases viability of lymphocytes in poultry.[21]
Humans and animals are constantly exposed to small concentrations of these mycotoxins, either individually or in combination.[22] Mycotoxicoses occur seasonally in areas that have not implemented effective prophylactic measures.[23] While interactions between mycotoxins had been discussed,[24] few studies have been conducted with these combinations.
Cochorus olitorius (C. olitorius, Tiliaceae family) is indigenous to the Middle East, including Egypt and South Africa. Young leaves of C. olitorius are regarded to be a healthy vegetable in East Asia and Japan, typically known as moroheiya.[25,26] Its health benefits have been reported to include antitumor activity by inhibiting tumorigenesis,[27] antioxidant properties,[28] and antibacterial activity.[29] Young leaves of C. olitorius are rich in calcium, potassium, phosphate, iron, ascorbic acid, carotene and other nutrients, and contain a large amount of mucilaginous polysaccharides.[28,30] It has also been reported that compounds such as carotenoids, flavonoids, and vitamin C, isolated from leaves of C. olitorius, exhibit significant antioxidant characteristics.[30] In addition, leaves of C. olitorius have been reported to have ethno-medicinal importance as a demulcent and febrifuge[31] and also possess anti-inflammatory, analgesic, and antimicrobial activities.[32,33] The aim of the current research was to assess possible protective effects of C. olitorius extracts against cytotoxic effects and disruption of DNA integrity induced by FB1 and AFB1 in the rat hepatoma cell line (H4IIE-luc).
Methods
Chemicals
Aflatoxin B1 and FB1 (98% purity) were purchased from Sigma Chemicals (St. Louis, MO, USA). The DNA extraction kit (DNeasy Blood and Tissue Kit) was obtained from Qiagen (Hilden, Germany). A DNA ladder, polymerase chain reaction (PCR) master mix containing 100 base pairs and RNAse free water were obtained from Fermentas Inc., (Glen Burnie, MD, USA). Supertherm Taq polymerase was purchased from JMR Holdings (London, UK). Forty primers were obtained from Operon Technologies (Alameda, CA, USA). All solvents used were analytical grade from Burdick and Jackson (Muskegon, MI, USA).
Plant materials
Stems and leaves of C. olitorius were collected from a residential garden in the city of Potchefstroom, North West Province, South Africa. The plant material was freeze-dried, pulverized, and 1 g was infused with 10 mL water for 24 h at room temperature. After centrifugation, the supernatant was freeze-dried and stored at 4 °C until used.
Cytotoxicity
Rat hepatoma cells (H4IIE-luc) were used as the mammalian model. This cell line had been stably transfected with a firefly luciferase reporter gene under control of the dioxin response element and the aryl hydrocarbon receptor mechanism.[34-37] These cells were originally developed as a reporter gene assay to determine the presence of, and to semi-quantify the concentrations of certain groups of persistent organic pollutants[38] including mixtures.[39]
H4IIE-luc cells were seeded at a density of 10,000 cells/mL media (Dulbecco’s Modified Eagle’s Medium, Sigma: D2902; St. Louis, MO, USA) in the inner 60 wells of a 96-well microplate. A volume of 250 μL of culture medium, supplemented with 0.044 mol/L NaHCO3 and 10% fetal bovine serum (Life Technologies, Carlsbad, CA, USA), was added to each well. To avoid edge effects and to create a homogenous microclimate across all wells containing cells, outer cells received 250 μL Dulbecco's phosphate buffered saline (PBS) (Tewksbury, MA, USA). Two sets of plates were incubated, at 37 °C in humidified air with 5% CO2, with one set for 24 h and the other set for 48 h. After incubation, the medium was removed and replaced with medium containing C.olitorius extract at either of two concentrations (20 or 40 μg/mL) and incubated for another 24 h. The medium was replaced with medium containing varying concentrations of AFB1 (50, 25, 2.5, 0.25, 0.025 μmol/L) dissolved in methanol,or of FB1 (200, 100, 10, 1, 0.1 μmol/L) dissolved in methanol. A combination of the already mentioned concentrations of AFB1 and FB1 were also tested: 50 μmol/L AFB1 +200 μmol/L FB1; 25 μmol/L AFB1+100 μmol/L FB1 , and so on. Exposureto mycotoxins was carried out in triplicates. Cells in six wells in each plate were exposed only to the aqueous extract of C. olitorius and cells in 11 wells were not exposed to anything except the growth medium.
To determine the viability, based on metabolic activity of cells, a colorimetric assay was performed using the yellow dye 3-(4,5-dimethyltiazol-2yl)-2,5-diphenyl tetrazolium bromide (MTT, Montigny-le-Bretonneux, France). In this assay, MTT is converted to formazan (blue) by mitochondrial reductase enzymes in living cells.[40] A final concentration of 500 μg/mL MTT was added to each well and incubated for 30 min. Blue formazan crystals that were formed by reduced MTT were dissolved with dimethylsulfoxide and absorbance by the formazan was measured spectrophotometrically at 560 nm. The amount of blue formazan produced is proportional to the amount of viable cells, and the percentage of viable to dead cells was calculated by comparison with a control (untreated and solvent control). Viability among various C. olitorius treatments described above were compared to the viability of cells treated only with mycotoxins by applying the same protocol described before, but omitting aqueous extract of C. olitorius.
Extraction of DNA
Harvested cells were washed with PBS to remove the nonadherent dead cells. The adherent cells were removed by trypsinizing (0.25% trypsin, 0.1% versene EDTA; purchased from Thermo Scientific, Rockford, IL, USA) and activity was stopped by addition of media. The cell suspension was centrifuged at 3,000 g for 5 min at room temperature. Genomic DNA was extracted from cells according to the Qiagen instruction manual and concentrations determined spectrophotometrically by use of the NanoDrop ND-1,000 Spectrophotometer. Purity of DNA was assessed by examining the 260/280 nm ratio.[41]
Random amplification of polymorphic DNA-polymerase chain reaction analysis
Amplification of DNA fragments was carried out using an ICycler (Bio-Rad, Herts, UK) thermal cycler using 20 primers from the Operon Biotechnologies (BioCampus Colonge Nattermannalle, Germany). PCR amplification was conducted in 25 μL reaction volumes containing 10 ng genomic DNA, 12.5 pmol/L master mix (×2) (Thermo Fisher Scientific, Carlsbad, CA, USA), 1.0 units of Supertherm Taq polymerase and 50 pmol/L primer. The PCR reactions were carried out in a thermocycler (Bio-Rad C1000, Bio-Rad, Hercules, CA, USA), programed with a first denaturation for 5 min at 95 °C, followed by 40 cycles for 30 s denaturation at 95 °C, 30 s annealing at 37 °C and 1 min extension at 72 °C. Final extension at 72 °C for 5 min was allowed before holding at 4 °C for 5 min. Reaction products were stored at -80 °C prior to electrophoresis.
Gel electrophoresis
Amplified products together with marker (100 bp DNA) were resolved by gel electrophoresis (60 V/cm for 135 min) on 2% agarose gel in tris-acetate-EDTA buffer containing 0.001 mg/mL ethidium bromide purchased from Sigma Chemical Co. (St. Louis, MO, USA). Gels were photographed by Gel Documentation System (Gensnap) software (Synegen, UK).
Band analysis
The gels for control and exposed DNA were run for each of the 20 primers [Table 1]. A DNA ladder of 100 bp was also run in each gel. The bands for PCR products were analyzed by TotalLab Quant (V11.5: TL100-LX59-7YF4-EX). The fluorimetric profiles of each amplification reaction were studied both qualitatively and quantitatively by comparing profiles from control and DNA exposed to the extracts. Each change observed in random amplification of polymorphic DNA (RAPD) profiles of treated groups (disappearances and appearance of bands in comparison to the control RAPD profiles) was given the arbitrary score of +1. The mean was then calculated for each experimental group exposed to the mycotoxins for varying time periods. Template genomic stability (%) was calculated as "100 - (100a/n)" where "a" is the average number of changes in DNA profiles and "n" is the number of bands selected in control DNA profiles.[42]
Sequences of the primers used to amplify DNA of H4IIE-luc rat hepatoma cells
| Primer | Sequence 5’-3’ | Primer | Sequence 5’-3’ |
|---|---|---|---|
| D01 | ACCGCGAAGG | D11 | AGCGCCATTG |
| D02 | GGACCCAACC | D12 | CACCGTATCC |
| D03 | GTCGCCGTCA | D13 | GGGGTGACGA |
| D04 | TCTGGTGAGG | D14 | CTTCCCCAAG |
| D05 | TGAGCGGACA | D15 | CATCCGTGCT |
| D06 | ACCTGAACGG | D16 | AGGGCGTAAG |
| D07 | TTGGCACGGG | D17 | TTTCCCACGG |
| D08 | GTGTGCCCCA | D18 | GAGAGCCAAC |
| D09 | CTCTGGAGAC | D19 | CTGGGGACTT |
| D10 | GGTCTACACC | D20 | ACCCGGTCAC |
Statistical analysis
All data were statistically analyzed with the Graphpad Prism 4.02 Inc. (La Jolla, CA, USA). The significance of the differences among treatment groups was determined with two-way analysis of variance. The assumptions of parametric statistics were confirmed. Normality was confirmed by the Kolmogorov-Smirnov test and homogeneity of variance was confirmed by use of Levine’s test. All statements of significance were based on a probability of P ≤ 0.05.
Results
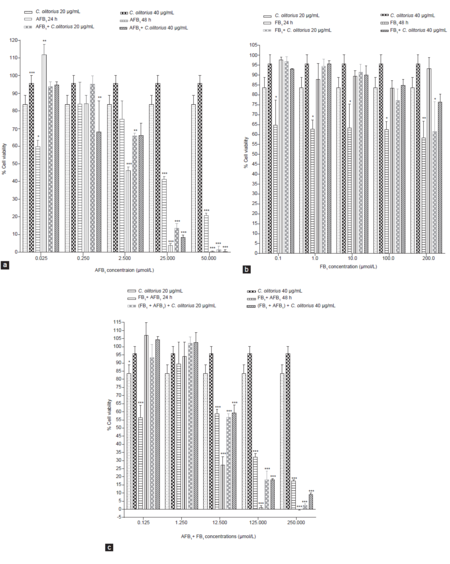
The results of cell viability assay revealed that H4IIE-luc cells that were treated with both concentrations (20 and 40 μg/mL) of C. olitorius extract were statistically significantly more viable after 24 h exposure than the ones that were not treated with the plant extract. However, there was no significant protection by the plant extract against FB1 after 48 h of exposure. Furthermore, C. olitorius extract could not protect the cells from the AFB1 concentration series or the combination exposure (AFB1 + FB1) irrespective of the exposure period (24 or 48 h) [Table 2]. A dose-dependent decrease of cell viability after exposure to increasing amounts of AFB1 was observed only after 48 h [Figure 1a]. After 24 h exposure, the response was not linear (hormetic effect). Except for the lowest concentration, cytotoxicity was more pronounced after 48 h. However, protective effects of the C. olitorius extract were observed after both 24 h and 48 h of exposure to AFB1. After 48 h, viability, expressed as a percentage of H4IIE-luc cells affected by FB1, was approximately 40% less than that of cells exposed to FB1 alone. After 48 h, there was also no dose-dependence, but cytotoxicity was less pronounced. Protective effects of 20 or 40 μg/mL C. olitorius extract were observed. After both 24 and 48 h of exposure, production of MTT formazan was greater in the presence of both concentrations of C. olitorius extract at all tested doses of FB1 compared to those in the absence of C. olitorius extract [Figure 1b]. No significant differences were found between 20 and 40 μg/mL of C. olitorius extract after 24 h of exposure.
Summary of Wilcoxon matched pair tests to compare the viability of rat hepatoma H4IIE-luc cell line treated with C. olitorius extract
| Mycotoxins | Exposure time | C. olitorius extract concentrations | |
|---|---|---|---|
| 20 μg/mL | 40 μg/mL | ||
| FB1 | 24 h | 0.04* | 0.04* |
| 48 h | 0.69 | 0.89 | |
| AFB1 | 24 h | 0.9 | 0.5 |
| 48 h | 0.35 | 0.89 | |
| FB1 + AFB1 | 24 h | 0.69 | 0.5 |
| 48 h | 0.22 | 0.08 | |
Figure 1. Cytotoxcity of (a) AFB1 at concentrations of 0.25-50 μmol/L without and with C. olitorius extract, (b) FB1 at concentration of 1-200 μmol/L without and with C. olitorius extract, and (c) AFB1 at concentrations of 0.25-50 μmol/L together with concentrations of 1-200 μmol/L FB1, on proliferation of H4IIE-luc cell line determined by MTT bioassay. Data represent mean ± SEM of triplicates (signifi cance of the differences among treatment groups: *P < 0.05; **P < 0.01; ***P < 0.001). AFB1: aflatoxin B1; FB1: fumonisin B1; C. olitorius: Cochorus olitorius; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; SEM: standard error mean
Incubation of H4IIE-luc cells with AFB1 + FB1 for 24 h resulted in greater cytotoxicity to cells as measured by the MTT assay, with significant toxicity at the sum of the two mycotoxin concentrations 12.5 and 125 μmol/L [Figure 1c]. The cells were least viable when they were exposed to the mixture of 250 μmol/L mycotoxin. Addition of C. olitorius extract to cells resulted in slightly greater viability. At lesser concentrations of AFB1 (1.25 μmol/L) + FB1 (12.5 μmol/L), protective ffects of aqueous extracts of C. olitorius on viability of cells was greater relative to the cells that did not receive plant extract [Figure 2].
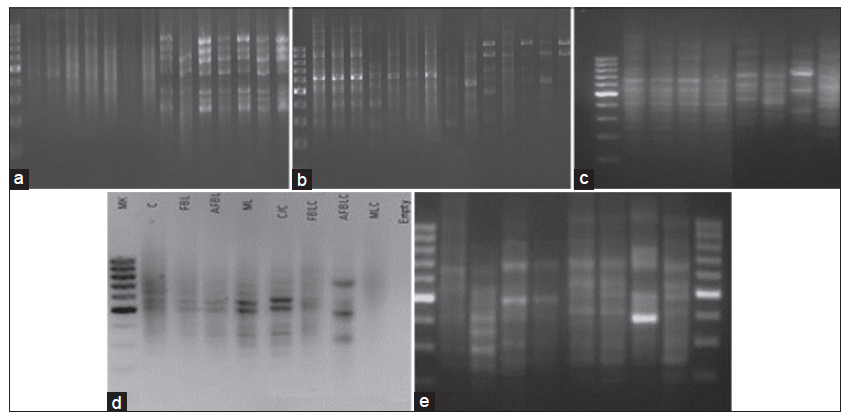
Figure 2. RAPD profi les of genomic DNA from cell line of rat, hepatoma (H4IIE-luc) cells, following exposure to FB1 and/or AFB1 for various time periods. (a) PCR products with primer OPD 07. (b) PCR products with primer OPD 09. Lane 1: the DNA marker (100 pb); lane 2: cells only; lane 3: cells plus FB1 (1 μmol/L); lane 4: cells plus FB1 (200 μmol/L); lane 5: cells plus AFB1 (0.25 μmol/L); lane 6: cells plus AFB1 (50 μmol/L); lane 7: cells plus mixture (1 μmol/L FB1 + 0.25 μmol/L AFB1); lane 8: cells plus mixture (200 μmol/L FB1 + 50 μmol/L AFB1); lane 9: cells plus C. olitorius (40 μg/mL); lane 10: C. olitorius (40 μg/mL) plus FB1 (1 μmol/L); lane 11: C. olitorius (40 μg/mL) plus FB1 (200 μmol/L); lane 12: C. olitorius (40 μg/mL) plus AFB1 (0.25 μmol/L); lane 13: C. olitorius (40 μg/mL) plus AFB1 (50 μmol/L); lane 14: C. olitorius (40 μg/mL) plus (1 μmol/L FB1 + 0.25 μmol/L AFB1); and lane 15: C. olitorius (40 μg/mL) plus (200 μmol/L FB1 + 50 μmol/L AFB1). (c) PCR products with primer OPD 13. (d) PCR products with primer OPD 16. Lane 1: DNA marker (100 pb); lane 2: cells only; lane 3: cells plus FB1 (1 μmol/L); lane 4: cells plus AFB1 (0.25 μmol/L); lane 5: cells plus mixture (1 μmol/L FB1 and 0.25 μmol/L AFB1); lane 6: cells plus C. olitorius (40 μg/mL); lane 7: C. olitorius (40 μg/mL) plus FB1 (1 μmol/L); lane 8: C. olitorius (40 μg/mL) plus AFB1 (0.25 μmol/L); and lane 9: C. olitorius (40 μg/mL) plus mixture (1 μmol/L FB1 and 0.25 μmol/L AFB1). (e) PCR products with primer OPD 16. Lane 1 and 10: DNA marker (100 pb); lane 2: cells only; lane 3: cells plus FB1 (200 μmol/L); lane 4: cells plus AFB1 (50 μmol/L); lane 5: cells plus mixture (200 μmol/L FB1 and 50 μmol/L AFB1); lane 6: cells plus C. olitorius (40 μg/mL); lane 7: C. olitorius (40 μg/mL) plus FB1 (200 μmol/L); lane 8: C. olitorius (40 μg/mL) plus AFB1 (50 μmol/L); and lane 9: C. olitorius (40 μg/mL) plus mixture (200 μmol/L FB1 and 50 μmol/L AFB1). AFB1: aflatoxin B1; FB1: fumonisin B1; C. olitorius: Cochorus olitorius; RAPD: random amplifi cation of polymorphic DNA; PCR: polymerase chain reaction. (a) OPD 07 for lesser (24 h) exposure; (b) OPD 09 for greater (48 h) exposure; (c) OPD 13 for greater (48 h) exposure; (d) OPD 16 for lesser (24 h) exposure; (e) OPD 16 for greater (48 h) exposure
The EC50 values for AFB1 were 6.9 and 1.8 after 24 and 48 h of exposure, respectively. When C. olitorius extract was added, the EC50 values were 4.3 and 2.49 after 24 or 48 h of exposure, respectively [Table 3]. At the lesser concentration, FB1 did not cause measurable cytotoxicity. However, the MTT assay revealed cytotoxicity at the greater concentration (200 μmol/L) although all doses studied were less than those required to obtain an EC50.
EC50 values of AFB1, FB1, and AFB1+ FB1 alone or in combination with the C. olitorius extract after 24 and 48 h and exposure measured by the MTT bioassay using H4IIE-luc rat hepatoma cells
| Mycotoxin and/or plant extract treatments | Time exposure (h) | Cytotoxicity (EC50) H4IIE-luc |
|---|---|---|
| FB1 | 24 | ND |
| 48 | ND | |
| AFB1 | 24 | 6.90 |
| 48 | 1.95 | |
| FB1 + AFB1 | 24 | 14.5 |
| 48 | 6.8 | |
| FB1 + C. olitorius (20 μg/mL) | 24 | 542.8 |
| FB1 + C. olitorius (40 μg/mL) | 24 | 26646 |
| AFB1 + C. olitorius (20 μg/mL) | 24 | 4.32 |
| AFB1 + C. olitorius (40 μg/mL) | 24 | 2.42 |
| FB1 + AFB1 + C. olitorius (20 μg/mL) | 24 | 18.5 |
| FB1 + AFB1 + C. olitorius (40 μg/mL) | 24 | 21.77 |
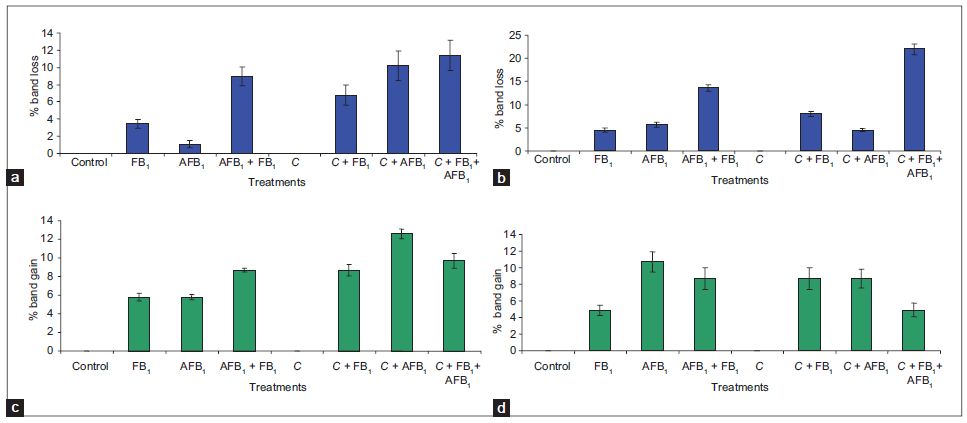
Only 5 of 10 oligonucleotide primers, primers D07, D09, D13, D15, and D16, used to measure responses of molecular-genetic parameters of cells among various treatments, gave detectable bands [Figure 3]. A total of 75 DNA sequences, ranging from 144 to 2,000 bp, were observed. All of the bands were “polymorphic” given 100% polymorphism for control cells and the other treatments for the 2 time periods using all primers. Quantitative analysis of these bands, expressed as a percentage of band loss, showed a time-dependent relationship [Figure 3 and Table 4]. Similarly, in the case of losses of bands after the shorter period of exposure (24 h), 12 of 75 bands (16%) had disappeared [Figure 3a]. At the longer duration of exposure (48 h), 21 of 75 bands (28%) had disappeared [Figure 3b]. Protective effects of C. olitorius extract were observed after 24 h, when 25 of 75 bands (33.3%) had disappeared, while for the 48 h and exposure, 30 of 75 bands (40%) had disappeared.
Figure 3. Genomic damage: the percentage of altered bands in each treatment at high concentration detected by RAPD-PCR. (a) Average band loss at lesser concentrations for 24 h; (b) average band loss at greater concentrations for 48 h; (c) average band gains at lesser concentrations for 24 h; and (d) average band gain at greater concentrations for 48 h. AFB1: aflatoxin B1; FB1: fumonisin B1; C: Cochorus olitorius; RAPD: random amplifi cation of polymorphic DNA; PCR: polymerase chain reaction
In cases where bands were gained after exposure to C. olitorius extract at the shorter duration of exposure, 21 new bands out of 75 (28%) were amplified. A similar trend was observed during the longer exposure, where 25 of 75 bands (33.3%) appeared [Figure 3c]. Protective effects of C. olitorius extract were observed as new bands appeared during the 24 h, since 32 of 75 bands (42.7%) appeared; during the longer exposure, 23 of 75 bands (30.7%) disappeared [Figure 3d].
When OPD 9 primer was used, a maximum of 10 RAPD-PCR disappeared when cells were exposed to the mixture of FB1 and AFB1 + aqueous extract of C. olitorius for 48 h [Table 4]. However, when with OPD 15 was used as the primer, the maximum appearance of new bands showed the same number of bands lost (10) that was observed in cells exposed to AFB1 + aqueous extract of C. olitorius after 24 h.
Frequency of appearance and disappearance of bands in the RAPD profiles of genomic DNA from H4IIE-luc rat hepatoma cell line following exposure to FB1 and/or AFB1 alone and in combination with the C. olitorius extract for 24 and 48 h
| Primer | Change in the RAPD profile | T1 | T2 | T3 | T4 | T5 | T6 | T7 | T8 | T9 | T10 | T11 | T12 | T13 | T14 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OPD 7 (24 h) | Appeared | 0 | 3 | 5 | 4 | 4 | 0 | 6 | 0 | 0 | 1 | 1 | 1 | 3 | 0 |
| Disappeared | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | |
| OPD 9 (48 h) | Appeared | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Disappeared | 0 | 0 | 0 | 1 | 3 | 3 | 2 | 0 | 0 | 1 | 5 | 0 | 4 | 10 | |
| OPD 13 (48 h) | Appeared | 0 | 0 | 0 | 0 | 3 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Disappeared | 0 | 0 | 3 | 0 | 0 | 0 | 2 | 0 | 4 | 6 | 4 | 4 | 6 | 5 | |
| OPD 15 (24 h) | Appeared | 0 | 0 | 0 | 1 | 4 | 6 | 3 | 0 | 5 | 2 | 10 | 3 | 0 | 0 |
| Disappeared | 0 | 3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 4 | |
| OPD 16 (48 h) | Appeared | 0 | 2 | 0 | 1 | 0 | 0 | 0 | 0 | 4 | 6 | 3 | 6 | 7 | 5 |
| Disappeared | 0 | 0 | 0 | 0 | 2 | 5 | 8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
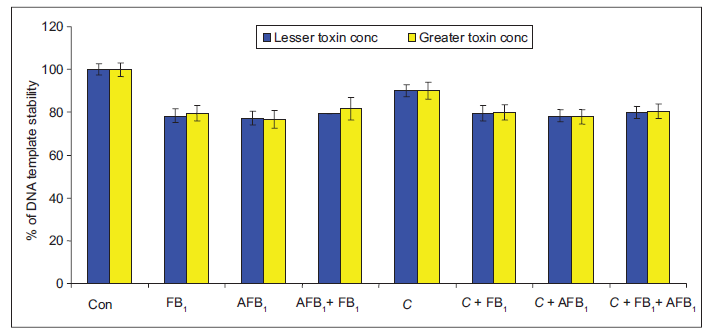
There was a significant difference in stability of the DNA template between control and each of the treated groups [Figure 4]. However, no significant difference was observed in stability of the DNA template between control and cells exposed to the aqueous extract of C. olitorius alone. The protective effect of the aqueous extract of C. olitorius on DNA was observed in the cells exposed to FB1 and AFB1.
Figure 4. Stability (%) of DNA templates, as determined RAPD-PCR in rat hepatoma cells (H4IIE-luc) following exposure to FB1 and/or AFB1 for 24 or 48 h. Con: control; AFB1: aflatoxin B1; FB1: fumonisin B1; C: Cochorus olitorius; RAPD: random amplification of polymorphic DNA; PCR: polymerase chain reaction
Discussion
Aflatoxin B1 and FB1 are the most frequently observed mycotoxins in food and animal feed. In African and European countries, both mycotoxins are found in maize.[43] Toxicity and carcinogenicity of AFB1, which has been classified as Group 1 carcinogen are thought to be directly linked to its bioactivation, resulting in a reactive form of AFB1, the 8, 9-epoxide. Bioactivation of AFB1 occurs primarily by a microsomal cytochrome P450-dependent epoxidation of the terminal furan ring of AFB1 , which is responsible for binding to cellular macromolecules such as DNA, RNA and other protein constituents.[44-47] The MTT assay is more sensitive and reproducible than testing intact animals and is valuable in determining the modes of action of toxins. In the current study, H4IIE-luc cells responded to FB1 and AFB1 as well as a mixture of the two mycotoxins. Cytotoxic effects of FB1 have been previously observed for murine microglial cells and primary astrocytes,[48] rat glioblastoma cells,[49,50] human keratinocytes and esophageal epithelial cells,[51] primary rat hepatocytes and rat liver in vivo.[52] In human and rat glioblastoma cells and mouse hypothalamic cells, production of ROS was increased after exposure to 10-100 μmol/L of FB1for 48-144 h.[53] Exposure to 10-100 μmol/L of FB1 for 72 h had no effect on production of ROS in human fibroblasts, or in primary cultures of rat astrocytes exposed to the same concentrations of FB1 for as long as 6 days.[54,55] Exposure to concentrations as high as 20 μmol/L of FB1 did not significantly reduce the viability of IPEC-J2 cells.[56]
In the current study, EC50 could not be calculated for FB1 because viability of cells exposed to 200 μmol/L was reduced only 41.6%, which is consistent with previously published results.[44] In yet another study, FB1 was only weakly cytotoxicity.[57] The EC50 for AFB1 was 1.87 μmol/L, which is similar to that observed previously by others,[58-60] who reported EC50 values ranging from 0.065 μmol/L forB-CMV1A2 cells to 14 μmol/L in BE12-6 cells. Exposure of H4IIE-luc cells to greater concentrations of AFB1 and FB1 resulted in lethality that was a concentration- and time-dependent. This effect was greater in cells treated with AFB1 or AFB1 + FB1. The interaction of FB1 and AFB1 in the induction of DNA damage and its correlation with biomarkers of cellular oxidative status has previously been reported to occur in vivo.[4,8,22,61] These reports suggested that genotoxicity and carcinogenicity of AFB1 were enhanced by exposure to FB1.[8] The in vivo results indicated that these effects were due to the production of ROS, which resulted in lipid peroxidation.[4,61]
AFB1 is a well-known genotoxicant. When the mechanism by which the aqueous extract of C. olitorius protected H4IIE-luc rat hepatoma cells against genetic damage caused by AFB1 and/or FB1 was investigated by use of RAPD analysis, there were statistically significant differences in the profiles of expression of the investigated genes, between the control and the treated cell lines at all concentrations tested.[33] Differences in the profile between the control and the treated samples were due to point mutations and/or base modifications of the genome caused by AFB1 and/or FB1.[62] Changes were observed for all genes for which primers were used. In our study, both qualitative and quantitative analyses showed that both mycotoxins increased instability of DNA templates of cells, in time- and concentration-dependent manners. This result supports the conclusion that both mycotoxins are direct-acting, genotoxicants that have the potential to attack hotspots present in DNA. The number of stable bands increased as a function of time and dose. Inconsistency in profiles of bands in RAPD analyses might have been observed because the two mycotoxins are acting directly as genotoxicants. However, they might act as genotoxicants through generation of free radicals during metabolism of the toxins through reactions of either electrophiles or nucleophiles with DNA. This interaction creates changes in their sequences that ultimately results in the formation of new priming sites and/or disappearances of existing priming sites for the RAPD primers. Thus, it gives different RAPD profiles for cells exposed to toxins.[63]
Random amplification of polymorphic DNA-PCR suffers from inherent limitations such as a lack of reproducibility and occurrence of pseudo-bands which prevent its routine application.[64] However, if conditions of the assays are properly optimized, these limitations can be resolved.[65,66] By optimizing conditions of the analysis, cloning the PCR products and further sequencing the products, RAPD can be useful in analyzing the nature and mode of action of the genotoxicants.[65,66] While in the present study RAPD could detect toxin-induced DNA damage, further studies would be needed before it could be used regularly as a tool in the detection of alterations in DNA sequence due to the genotoxicants.
Previous studies have demonstrated that certain compounds in the diet can offer protection against toxicity of mycotoxins.[67] Natural vitamins, carotenoids, polyphenol and trace elements are potentially beneficial in protection against mycotoxicosis.[68] Green leafy vegetables are known to be dietary sources of minerals, trace elements and phytochemicals that contribute to health.[69] Molecular evidence has suggested that trace elements and antioxidant molecules in green, leafy vegetables lessen risks of cancer and cardiovascular diseases through mechanisms that modulate free radical attack on nucleic acids, proteins, and polyunsaturated fatty acids.[70]C. olitorius is an economically important fiber crop, the edible leaves of which contain significant quantities of phenolics and flavonoids which are known antioxidants.[33,71-74] Although in the current study the active compound(s) in the aqueous extract of C. olitorius were not isolated or identified, flavonoids are possible candidates among the active compound(s) in C. olitorius. C. olitorius contains abundant amounts of a number of flavonoids that could act as antioxidants, including: 5-caffeoylquinic acid, 3,5-dicaffeoylquinic acid, quercetin 3-galactoside, quercetin 3-glucoside, quercetin 3-(6-malonylglucoside), quercetin 3-(malonylgalactoside), ascorbic acid, a-tocopherol, and chlorophyll.[29] Furthermore, C. olitorius contains relatively high levels of quercetin glycosides. Several novel flavonol glycosides named corchorusides A and B, in addition to a major component, capsugenin-25, 30-O-β-diglucopyranoside have been isolated from C. olitorius.[26] Recently, several flavonoids, such as rutin, and quercetin and phenolic compounds, including gallic acid, chlorogenic acid, p-cumaric acid, ferulic acid, and ellagic acid have been isolated from extracts of C. olitorius.[75] Consequently, protective effects of the aqueous extract of C. olitorius against cytotoxicities of AFB1 and FB1 in H4IIE-luc might be due to the antioxidant capacity and the abundant occurrence of the flavonoid compounds. Another candidate for the active compound(s) is chlorophyll. Numerous in vitro studies have indicated that derivatives of chlorophyll, including chlorophyllide A and B and pheophorbide A and B can attenuate chemical genotoxicity by forming a molecular complex with pro-mutagens,[74-76] which might involve strong chlorophyll-AFB1 and/or FB1 interaction via their planar unsaturated cyclic rings.[75] Derivatives of pheophorbide A and B provided additional protection not only by direct trapping, but also by increasing glutathione S-transferase activity against hepatic AFB1 metabolites.[76]
In conclusion, both AFB1 and FB1 induced oxidative stress, which resulted in cytotoxicity and fragmentation of DNA of H4IIE-luc rat hepatoma cells after various durations of exposure to these toxins singly or in combination. Exposure to these mycotoxins resulted in appearance of new bands in the RAPD analysis, in addition to DNA damage. Treatment with an aqueous extract of C. olitorius resulted in a significant improvement in viability of cells and reduced damage to DNA in H4IIE-luc cells exposed to mycotoxins. Due to these effects, C. olitorius is suggested to be a traditional edible plant containing potential chemo-preventive agents for human cancers. However, additional studies on the uptake, metabolism, and disposition of the active ingredients in C. olitorius need to be further studied. Currently, the active ingredient (s) are unknown, and it is also not known whether these constituents that are effective in vitro can have similar effects in vivo.
Acknowledgments
The Morogo Research Program gratefully acknowledges the National Research Foundation of South Africa (Focus Area Grant FA2004050600064) and National Research Center, Cairo, Egypt Project #10070112 for financial support of this study. Prof. Giesy was supported by the Canada Research Chair program, a Visiting Distinguished Professorship in the Department of Biology and Chemistry and State Key Laboratory in Marine Pollution, City University of Hong Kong, the 2012 “High Level Foreign Experts” (#GDT20143200016) program, funded by the State Administration of Foreign Experts Affairs, the P.R. China to Nanjing University and the Einstein Professor Program of the Chinese Academy of Sciences.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
1. Ibá-ez-Vea M, González-Pe-as E, Lizarraga E, López de Cerain A. Co-occurrence of mycotoxins in Spanish barley: a statistical overview. Food Control 2012;28:295-8.
2. Fink-Gremmels J. Moulds and mycotoxins as undesirable substances in animal feeds In: Barug D, Bhatnagar HP, van Egmond JW, van der Kamp WA, van Osenbruggen VA, editors. The mycotoxin fact book. Food and feed topics. Wageningen: Wageningen Academic Publishers; 1996. pp. 37-50.
3. Bhat R, Rai RV, Karim AA. Mycotoxins in food and feed: present status and future concerns. Compr Rev Food Sci Food Saf 2010;9:57-81.
4. Abdel-Wahhab MA, Ibrahim AA, El-Nekeety AA, Hassan NS, Mohamed AA. Panax ginseng C.A. Meyer extract counteracts the oxidative stress in rats fed multi-mycotoxins-contaminated diet. Com Sci 2012;3:143-53.
5. Potential economic costs of mycotoxins in United States. In: Mycotoxins: risks in plant, animal and human Task force reports. Ames, Iowa: Council for Agricultural Science and Technology; 2003. pp. 136-42.
6. Nogueira JA, Ono-Nita SK, Nita ME, de Souza MM, do Carmo EP, Mello ES, Scapulatempo C, Paranaguá-Vezozzo DC, Carrilho FJ, Alves VA. 249 TP53 mutation has high prevalence and is correlated with larger and poorly differentiated HCC in Brazilian patients. BMC Cancer 2009;9:204.
7. Abdel-Wahhab MA, Ahmed HH, Hagazi MM. Prevention of aflatoxin B1-initiated hepatotoxicity in rat by marine algae extracts. J Appl Toxicol 2006;26:229-38.
8. Abdel-Wahhab MA, Hassan NS, El-Kady AA, Khadrawy YA, El-Nekeety AA, Mohamed SR, Sharaf HA, Mannaa FA. Red ginseng extract protects against afl atoxin B1 and fumonisins-induced hepatic pre-cancerous lesions in rats. Food Chem Toxicol 2010;48:733-42.
10. International Programme on Chemical Safety. Aflatoxins. Available from: http://www.inchem.org/documents/jecfa/jecmono/v040je16. htm.[Last accessed on 2012 Jan 03].
11. Abdel-Wahhab MA, Nada SA, Farag IM, Abbas NF, Amra HA. Potential protective effect of HSCAS and bentonite against dietary aflatoxicosis in rat: with special reference to chromosomal aberrations. Nat Toxins 1998;6:211-8.
12. Abdel-Wahhab MA, Nada SA, Amra HA. Effect of aluminosilicates and bentonite on aflatoxin-induced developmental toxicity in rat. J Appl Toxicol 1999;19:199-204.
13. Williams JH, Phillips TD, Jolly PE, Stiles JK, Jolly CM, Aggarwal D. Human aflatoxicosis in developing countries: a review of toxicology, exposure, potential health consequences, and interventions. Am J Clin Nutr 2004;80:1106-22.
14. Matur E, Ergul E, Akyazi I, Eraslan E, Inal G, Bilgic S, Demircan H. Effects of Saccharomyces cerevisiae extract on haematological parameters, immune function and the antioxidant defence system in breeder hens fed aflatoxin contaminated diets. Br Poult Sci 2011;52:541-50.
15. Adedara IA, Owumi SE, Uwaifo AO, Farombi EO. Aflatoxin B1 and ethanol co-exposure induces hepatic oxidative damage in mice. Toxicol Ind Health 2010;26:717-24.
16. El-Nekeety AA, Mohamed SR, Hathout AS, Hassan NS, Aly SE, Abdel-Wahhab MA. Antioxidant properties of Thymus vulgaris oil against aflatoxin-induce oxidative stress in male rats. Toxicon 2011;57:984-91.
17. van der Westhuizen L, Shephard GS, Rheeder JP, Burger HM. Individual fumonisin exposure and sphingoid base levels in rural populations consuming maize in South Africa. Food Chem Toxicol 2010;48:1698-703.
18. Hassan AM, Mohamed SR, El-Nekeety AA, Hassan NS, Abdel-Wahhab MA. Aquilegia vulgaris L. extract counteracts oxidative stress and cytotoxicity of fumonisin in rats. Toxicon 2010;56:8-18.
19. International Programme on Chemical Safety. Fumonisin B1 Environmental Health Criteria 219. Available from: http://www.inchem. org/documents/ehc/ehc/ehc219.htm.[Last accessed on 2011 Oct 28].
20. Toxins derived from Fusarium moniliforme: fumonisins B1 and B2 and fusarin C. IARC Monogr Eval Carcinog Risks Hum 1993;56:445-66.
21. Dombrink-Kurtzman MA, Javed T, Bennett GA, Richard JL, Cote LM, Buck WB. Lymphocyte cytotoxicity and erythrocytic abnormalities induced in broiler chicks by fumonisins B1 and B2 and moniliformin from Fusarium proliferatum. Mycopathologia 1993;124:47-54.
22. Theumer MG, Cánepa MC, López AG, Mary VS, Dambolena JS, Rubinstein HR. Subchronic mycotoxicoses in Wistar rats: assessment of the
23. Pfohl-Leszkowicz A, Petkova-Bocharova T, Chernozemsky IN, Castegnaro M. Balkan endemic nephropathy and associated urinary tract tumours: a review on aetiological causes and the potential role of mycotoxins. Food Addit Contam 2002;19:282-302.
24. Pfohl-Leszkowicz A, Manderville RA. Ochratoxin A: an overview on toxicity and carcinogenicity in animals and humans. Mol Nutr Food Res 2007;51:61-99.
25. van der Walt AM, Ibrahim MI, Bezuidenhout CC, Loots du T. Linolenic acid and folate in wild-growing African dark leafy vegetables (morogo). Public Health Nutr 2009;12:525-30.
26. Phuwapraisirisan P, Puksasook T, Kokpol U, Suwanborirux K. Corchorusides A and B, new flavonol glycosides as a-glucosidase inhibitors from the leaves of
27. Furumoto T, Wang R, Okazaki K, Hasan AF, Ali MI, Kondo A, Fukui H. Antitumor promoters in leaves of jute (Corchorus capsularis and
28. Azuma K, Nakayama M, Koshioka M, Ippoushi K, Yamaguchi Y, Kohata K, Yamauchi Y, Ito H, Higashio H. Phenolic antioxidants from the leaves of
29. Zakaria ZA, Somchit MN, Zaiton H, Mat Jais AM, Sulaiman MR, Farah WO, Nazaratulmawarina R, Fatimah CA. The
30. Ohtani K, Okai K, Yamashita U, Yuasa I, Misaki A. Characterization of an acidic polysaccharide isolated from the leaves of
31. Nishiumi S, Yabushita Y, Fukuda I, Mukai R, Yoshida K, Ashida H. Molokhia (
32. Khan KS, Joergensen RG. Decomposition of heavy metal contaminated nettles (Urtica dioica L.) in soils subjected to heavy metal pollution by river sediments. Chemosphere 2006;65:981-7.
33. Das AK, Sahu R, Dua TK, Bag S, Gangopadhyay M, Sinha MK, Dewanjee S. Arsenic-induced myocardial injury: protective role of
34. Lee KT, Hong S, Lee JS, Chung KH, Hilscherová K, Giesy JP, Khim JS. Revised relative potency values for PCDDs, PCDFs, and non-ortho-substituted PCBs for the optimized H4IIE-
35. Murk AJ, Legler J, Denison MS, Giesy JP, van de Guchte C, Brouwer A. Chemical-activated luciferase gene expression (CALUX): a novel
36. Garrison PM, Tullis K, Aarts JM, Brouwer A, Giesy JP, Denison MS. Species-specific recombinant cell lines as bioassay systems for the detection of 2,3,7,8-tetrachlorodibenzo-p-dioxin-like chemicals. Fundam Appl Toxicol 1996;30:194-203.
37. Denison MA, El-Fouly MH, Aarts JM, Brouwer A, Richter C, Giesy JP. Production of novel recombinant cell line bioassay systems for detection of 2,3,7,8-tetrachlorodibenzo-p-dioxin-like chemicals In: Fiedler H, Frank H, Hutzinger O, Parzfall W, Riss A, Safe S, editors. Organohalogen compounds; emission control, transport and fate and environmental levels and ecotoxicology. Austria: Federal Environmental Agency; 1993. pp. 365-8.
38. Nie M, Blankenship AL, Giesy JP. Interactions between aryl hydrocarbon receptor (AhR) and hypoxia signaling pathways. Environ Toxicol Pharmacol 2001;10:17-27.
39. Larsson M, Giesy JP, Engwall M. AhR-mediated activities of polycyclic aromatic compound (PAC) mixtures are predictable by the concept of concentration addition. Environ Int 2014;73:94-103.
40. Vistica DT, Skehan P, Scudiero D, Monks A, Pittman A, Boyd MR. Tetrazolium-based assays for cellular viability: a critical examination of selected parameters affecting formazan production. Cancer Res 1991;51:2515-20.
41. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248-54.
42. Atienzar FA, Jha AN. The random amplified polymorphic DNA (RAPD) assay and related techniques applied to genotoxicity and carcinogenesis studies: a critical review. Mutat Res 2006;613:76-102.
43. Rafai P, Bata A, Jakab L, Ványi A. Evaluation of mycotoxin-contaminated cereals for their use in animal feeds in Hungary. Food Addit Contam 2000;17:799-808.
44. Kouadio JH, Mobio TA, Baudrimont I, Moukha S, Dano SD, Creppy EE. Comparative study of cytotoxicity and oxidative stress induced by deoxynivalenol, zearalenone or fumonisin B1 in human intestinal cell line Caco-2. Toxicology 2005;213:56-65.
45. Massey TE, Stewart RK, Daniels JM, Liu L. Biochemical and molecular aspects of mammalian susceptibility to aflatoxin B1 carcinogenicity. Proc Soc Exp Biol Med 1995;208:213-27.
47. Abdel-Wahhab MA, Omara EA, Abdel-Galil MM, Hassan NS, Nada SA, Saeed A, Saeed A, el-Sayed MM. Zizyphus spina-christi extract protects against aflatoxin B1 -initiated hepatic carcinogenicity. Afr J Tradit Complement Altern Med 2007;16:248-56.
48. Osuchowski MF, Sharma RP. Fumonisin B1 induces necrotic cell death in BV-2 cells and murine cultured astrocytes and is antiproliferative in BV-2 cells while N2A cells and primary cortical neurons are resistant. Neurotoxicology 2005;26:981-92.
49. Mobio TA, Anane R, Baudrimont I, Carratú MR, Shier TW, Dano SD, Ueno Y, Creppy EE. Epigenetic properties of fumonisin B1: cell cycle arrest and DNA base modification in C6 glioma cells. Toxicol Appl Pharmacol 2000;164:91-6.
50. Mobio TA, Tavan E, Baudrimont I, Anane R, Carratú MR, Sanni A, Gbeassor MF, Shier TW, Narbonne JF, Creppy EE. Comparative study of the toxic effects of fumonisin B1 in rat C6 glioma cells and p53-null mouse embryo fibroblasts. Toxicology 2003;183:65-75.
51. Tolleson WH, Melchior WB Jr, Morris SM, McGarrity LJ, Domon OE, Muskhelishvili L, James SJ, Howard PC. Apoptotic and anti-proliferative effects of fumonisin B1 in human keratinocytes, fibroblasts, esophageal epithelial cells and hepatoma cells. Carcinogenesis 1996;17:239-49.
52. Abel S, Gelderblom WC. Oxidative damage and fumonisin B1-induced toxicity in primary rat hepatocytes and rat liver
53. Stockmann-Juvala H, Mikkola J, Naarala J, Loikkanen J, Elovaara E, Savolainen K. Fumonisin B1-induced toxicity and oxidative damage in U-118MG glioblastoma cells. Toxicology 2004;202:173-83.
54. Galvano F, Campisi A, Russo A, Galvano G, Palumbo M, Renis M, Barcellona ML, Perez-Polo JR, Vanella A. DNA damage in astrocytes exposed to fumonisin B1. Neurochem Res 2002;27:345-51.
55. Galvano F, Russo A, Cardile V, Galvano G, Vanella A, Renis M. DNA damage in human fibroblasts exposed to fumonisin B1. Food Chem Toxicol 2002;40:25-31.
56. Wan LY, Turner PC, El-Nezami H. Individual and combined cytotoxic effects of Fusarium toxins (deoxynivalenol, nivalenol, zearalenone and fumonisins B1) on swine jejunal epithelial cells. Food Chem Toxicol 2013;57:276-83.
57. Kouadio JH, Dano SD, Moukha S, Mobio TA, Creppy EE. Effects of combinations of Fusarium mycotoxins on the inhibition of macromolecular synthesis, malondialdehyde levels, DNA methylation and fragmentation, and viability in Caco-2 cells. Toxicon 2007;49:306-17.
58. McKean C, Tang L, Tang M, Billam M, Wang Z, Theodorakis CW, Kendall RJ, Wang JS. Comparative acute and combinative toxicity of aflatoxin B1 and fumonisin B1 in animals and human cells. Food Chem Toxicol 2006;44:868-76.
59. Lewis CW, Smith JE, Anderson JG, Freshney RI. Increased cytotoxicity of food-borne mycotoxins toward human cell lines
60. Terse PS, Madhyastha MS, Zurovac O, Stringfellow D, Marquardt RR, Kemppainen BW. Comparison of
61. Theumer MG, López AG, Masih DT, Chulze SN, Rubinstein HR. Immunobiological effects of AFB1 and AFB1-FB1 mixture in experimental subchronic mycotoxicoses in rats. Toxicology 2003;186:159-70.
62. Singh KP, Roy D. Identification of novel breast tumor-specific mutation(s) in the q11.2 region of chromosome 17 by RAPD/AP-PCR fingerprinting. Gene 2001;269:33-43.
63. Bouhlel I, Limem I, Skandrani I, NefattiA, Ghedira K, Dijoux-Franca MG, Leila CG. Assessment of isorhamnetin 3-O-neohesperidoside from Acacia salicina: protective effects toward oxidation damage and genotoxicity induced by aflatoxin B1 and nifuroxazide. J Appl Toxicol 2010;30:551-8.
64. Hadrys H, Schierwater B, Dellaporta SL, DeSalle R, Buss LW. Determination of paternity in dragonflies by random amplified polymorphic DNA fingerprinting. Mol Ecol 1993;2:79-87.
65. Theodorakis CW, Bickham JW, Elbl T, Shugart LR, Chesser RK. Genetics of radionuclide contaminated mosquitofish populations and homology between Gambusia affinis and G. holbrooki. Environ Toxicol Chem 1998;17:1992-8.
66. Becerril C, Ferrero M, Sanz F, Casta-o A. Detection of mitomycin C-induced genetic damage in fish cells by use of RAPD. Mutagenesis 1999;14:449-56.
67. Rizzo AF, Atroshi F, Ahotupa M, Sankari S, Elovaara E. Protective effect of antioxidants against free radical-mediated lipid peroxidation induced by DON or T-2 toxin. J Vet Med Assoc 1994;41:81-90.
68. Galvano F, Piva A, Ritieni A, Galvano G. Dietary strategies to counteract the effects of mycotoxins: a review. J Food Prot 2001;64:120-31.
69. van der Walt AM, Loots DT, Ibrahim MIM, Bezuidenhout CC. Minerals, trace elements and antioxidant phytochemicals in wild African dark-green leafy vegetables (morogo). S Afr J Sci 2009;105:444-8.
70. Borek C. Cancer prevention by natural dietary antioxidants in developing countries In: Bahorun T, Gurib-Fakim A, editors. Molecular and Therapeutic Aspects of Redox Biochemistry. London: OICA International (U.K.) Limited; 2003. pp. 259-69.
71. Dewanjee S, Sahu R, Karmakar S, Gangopadhyay M. Toxic effects of lead exposure in Wistar rats: involvement of oxidative stress and the beneficial role of edible jute (
72. Negishi T, Arimoto S, Nishizaki C, Hayatsu H. Inhibitory effect of chlorophyll on the genotoxicity of 3-amino-1-methyl-5H-pyrido[4,3-b] indole (Trp-P-2). Carcinogenesis 1989;10:145-9.
73. Arimoto S, Fukuoka S, Itome C, Nakano H, Rai H, Hayatsu H. Binding of polycyclic planar mutagens to chlorophyllin resulting in inhibition of the mutagenic activity. Mutat Res 1993;287:293-305.
74. Tachino N, Guo D, Dashwood WM, Yamane S, Larsen R, Dashwood R. Mechanisms of the
75. Breinholt V, Schimerlik M, Dashwood R, Bailey G. Mechanisms of chlorophyllin anticarcinogenesis against aflatoxin B1: complex formation with the carcinogen. Chem Res Toxicol 1995;8:506-14.
Cite This Article
Export citation file: BibTeX | RIS
OAE Style
Ibrahim MIM, Pieters R, Abdel-Aziem SH, Walt AM, Bezuidenhout CC, Giesy JP, Abdel-Wahhab MA. Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells. Hepatoma Res 2015;1:75-85. http://dx.doi.org/10.4103/2394-5079.155692
AMA Style
Ibrahim MIM, Pieters R, Abdel-Aziem SH, Walt AM, Bezuidenhout CC, Giesy JP, Abdel-Wahhab MA. Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells. Hepatoma Research. 2015; 1: 75-85. http://dx.doi.org/10.4103/2394-5079.155692
Chicago/Turabian Style
Ibrahim, Mohamed I. M., Rialet Pieters, Sekena H. Abdel-Aziem, Anna M. van der Walt, Cornelius C. Bezuidenhout, John P. Giesy, Mosaad A. Abdel-Wahhab. 2015. "Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells" Hepatoma Research. 1: 75-85. http://dx.doi.org/10.4103/2394-5079.155692
ACS Style
Ibrahim, MIM.; Pieters R.; Abdel-Aziem SH.; Walt AM.; Bezuidenhout CC.; Giesy JP.; Abdel-Wahhab MA. Aqueous extract of Corchorus olitorius decreases cytotoxicity of aflatoxin B1 and fumonisin B1 in H4IIE-luc cells. Hepatoma. Res. 2015, 1, 75-85. http://dx.doi.org/10.4103/2394-5079.155692
About This Article
Copyright
Data & Comments
Data
 Cite This Article 1 clicks
Cite This Article 1 clicks












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.